
Science with Passion
Application No.: VSP0016
Version 1 02/2018
Quantification of caffeine with the AZURA® Educational system and Mobile Control Software
Juliane Böttcher, Mareike Margraf, Kate Monks; applications@knauer.net
KNAUER Wissenschaftliche Geräte GmbH, Hegauer Weg 38,
14163 Berlin

Summary
The AZURA® Educational system allows an easy and fast implementation of liquid chromatography (HPLC, high pressure liquid chromatography) and promotes a deeper understanding of this separation method. A simple example is given describing the determination of a sample containing caffeine and paracetamol.
Introduction
Caffeine and paracetamol are widely-used pharmaceutical components. Both substances are present as ingredients in many analgesics. Hence, they are often determined simultaneously in routine analysis. Theophylline, a substance chemically closely related to caffeine, is used to serve as an internal standard. [1] To analyze the components, the KNAUER HPLC Educational system is used providing isocratic elution HPLC in combination with UV detection. The samples are injected via a manual injection valve. Based on the KNAUER AZURA Compact series, this system layout represents an easy and convenient solution for the current application. The determination of a sample containing caffeine and paracetamol is a typical example from applied research for the implementation of the KNAUER HPLC Educational system.
Results
At first, stock solutions are prepared from caffeine, paracetamol and theophylline. The initial weight of the substances should be about 100 mg. However, it is important to note the exact sample weight to obtain accurate results for the quantitative analysis. All standards were dissolved and sonicated to yield stock solutions of approximately 10 mg/mL. To identify the individual substances directly by HPLC, the substances are diluted with water (Tab A1, Additional Materials and Methods). Secondly, a single calibration solution is prepared from the caffeine and paracetamol stock solution. For this purpose, 50 µL of the caffeine stock solution and 50 µL of the paracetamol stock solution are combined and diluted with water to a final volume of 5 mL. For the current application, five dilution levels (Tab A2, Additional Materials and Methods) have been prepared. Furthermore, 100 µL of the theophylline stock solution are diluted with water to a final volume of 1 mL. Subsequently, a volume of 20 µL of this solution is added to standard 1 – 5. Fig 1 shows the chromatogram of the calibration standard at level 4 (60µg/mL). The peaks are baseline separated in less than 5 minutes. Fig 2 shows the measurement of an analgesic sample. Therefore, analgesic tablets containing paracetamol and caffeine were chosen. The internal standard theophylline was added according to the preparation of calibration solutions. A concentration of 53 mg caffeine was calculated for the sample. This amount refers to the weight of one tablet. Relating to the package insert, the analgesic should contain 50 mg caffeine per tablet. The deviation of the measured and proclaimed value might result from differing calibrations and/or measurement errors. For the detailed preparation of standards, sample and calibration please see application VSP0018.
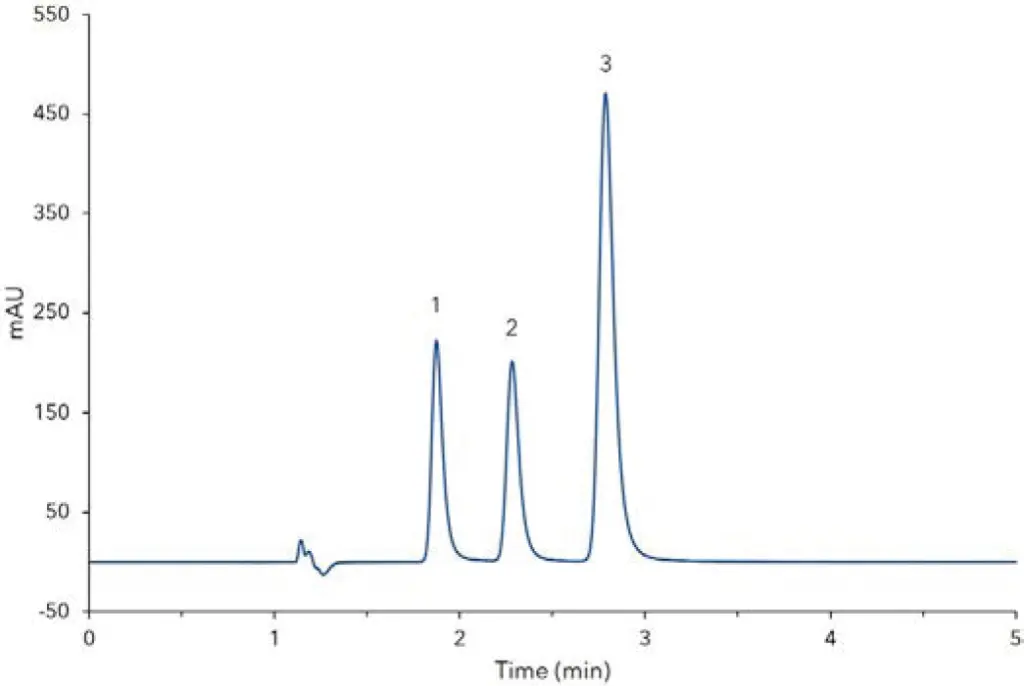
Fig. 1 Chromatogram of standard solution 4, 1) paracetamol, 2) theophylline (IS),3) caffeine
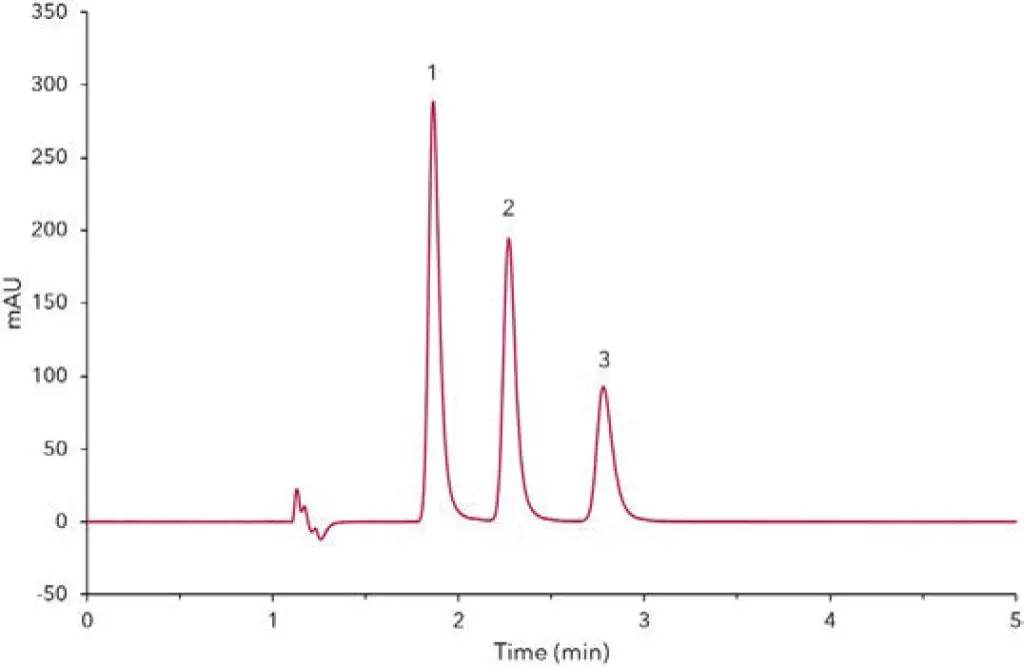
Fig. 2 Chromatogram of analgesic sample, 1) paracetamol, 2) theophylline (IS), 3) caffeine
Materials and Methods
For the determination of caffeine and paracetamol the AZURA Educational system was used, which combines a P 4.1S pump, an UVD 2.1S and a manual injection valve in just one AZURA assistant. The flow rate was set to 0.8 ml/min at ambient temperature. The wavelength was set to 273 nm with a data rate of 20 Hz and a time constant of 0.05 s. 10 µL of the standards and samples were injected. The isocratic method ran for 5 minutes with a mixture of methanol and water 40:60 (v/v). The column in a dimension 125 x 4 mm ID with precolumn was filled with Eurospher II 100-5 C18 silica. For the data acquisition the Mobile Control Chrom software was used.
Conclusion
The KNAUER HPLC Educational System provides both, a qualitative and quantitative analysis of caffeine from different chemical probes. The system is compact, very simple to operate and can be ideally used for practical training courses. The Mobile Control Chrom is an intuitive and cost-effective software solution for controlling and monitoring your AZURA devices and systems. In addition, you can acquire data from AZURA detectors allowing simple measurements.

Additional Results
Tab. A1 Initial weight and dilution of stock solutions
Tab. A2 Caffeine standards 1 to 5
Additional Materials and Methods
Tab. A3 Method parameters
Tab. A4 System configuration & data

AZURA® Educational system
References
[1] Entry: internal standard. In: IUPAC Compendium of Chemical Terminology (the “Gold Book”). doi:10.1351/goldbook.I03108.
Related KNAUER Applications
VSP0017 – Quantification of caffeine with the AZURA® Educational system and ClarityChrom software
VSP0018 – Preparation of calibration and samples for the quantification of caffeine with the AZURA® Educational system
VSP0019 – HPLC Basics – principles and parameters
Application details


