
Science with Passion
Application No.: VPH0064
Version 1 06/2019
Quality control of pharmaceutical solutions by determination of osmolality
Juliane Böttcher, Kate Monks; applications@knauer.net
KNAUER Wissenschaftliche Geräte GmbH, Hegauer Weg 38, 14163 Berlin

SUMMARY
The osmolality of solutions used in the clinical and pharmaceutical field is an important issue that requires regular monitoring. Especially infusion solutions but also solutions for external use like eye drops and rinsing solutions must be isotonic to ensure the physical well-being of humans. In this study, the osmolality of commercially available pharmaceutical solutions were determined using the KNAUER K-7400S Semi-Micro Osmometer.
INTRODUCTION
To guarantee the quality of solutions used for pharmaceutical or medical purposes the osmolality is consulted as an assessment value. The osmolality is a general measure for the number of solved molecules in a liquid and is commonly given in mOsmol/kg. Conventionally used solutions for clinical application are for example Ringer solution [1], physiological salt solution (0.9 % NaCl), and 5 % glucose. These have to be in the osmolality range of 290±10 mOsmol/kg to comply with human plasma [2]. In addition to these physiological infusions also glucose solutions of higher concentration (10 %, 15 % and 20 %) are used in daily clinical practice. These are for instance applied for the treatment of hypoglycemic conditions or as carbohydrate component in parenteral nutrition [3]. All of the mentioned solutions were prepared and analysed to evaluate their actual osmolalities.
RESULTS
To evaluate the results, they were divided into the different fields of application. On the one hand the isotonic solutions used for infusion or rinsing, and on the other hand the glucose solutions with varying concentrations for special treatments. The 5 % glucose solution is somehow an exception and could be applied for both ambits. The diagram in Fig 1 shows the measurement of the isotonic solutions in relation to the given limit value for plasma. The averaged osmolality (n=10) for Ringer solution was 284 mOsmol/kg, 281 mOsmol/kg for 0.9 % NaCl and 287 mOsmol/kg for 5 % glucose. The diagram in Fig 2 visualizes the measured osmolalities of different glucose solutions with concentrations of 5 %, 10 %, 15 % and 20 % glucose. The determined averaged osmolality (n=10) added up to 604 mOsmol/kg for 10 %.
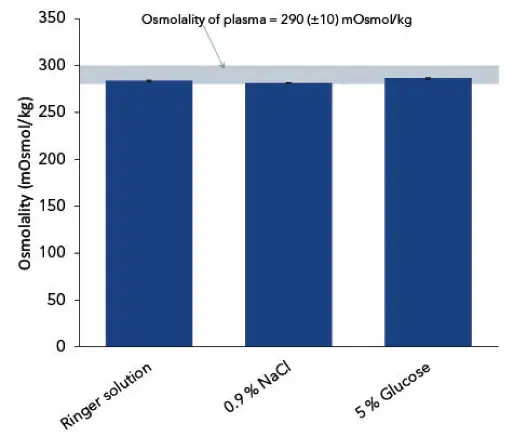
Fig. 1 Measured osmolalities of pharmaceutical infusion solutions. Graph shows average values and standard deviations of 10 replicates.
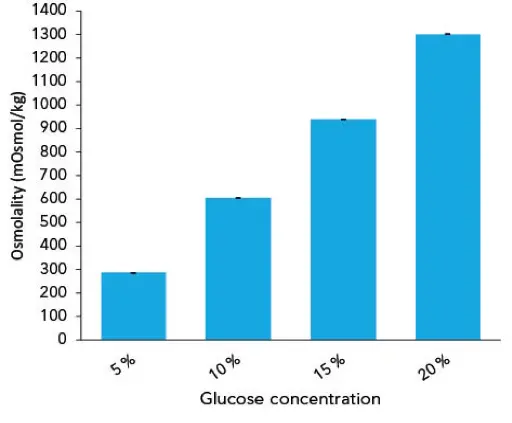
Fig. 2 Measured osmolalities of different glucose solutions.Graph shows average values and standard deviations of 10 replicates.
Materials and Methods
All measurements were made with the KNAUER K-7400S Semi-Micro Osmometer. The used calibration standards had osmolality values of 300, 400, 850, and 2000 mOsmol/kg. The system parameters were set to -8 °C for freeze and -16 °C for cooling limit. All prepared solutions were degassed using an ultrasonic bath to remove the carbon dioxide. Then 150 µL of the samples were transferred to a plastic sample tube.
Conclusion
The measured results for all isotonic solutions are within the given limit value for human plasma of 290±10 mOsmol/kg. This is interesting as most manufacturers of pharmaceutical solutions only state the theoretical osmolalities of their products. The theoretical osmolality of physiological salt solution is specified as 308 mOsmol/kg. However, as shown in the analysis, the real osmolality is clearly lower (281 mOsmol/kg). Especially for more complex infusion solutions, a verification of the real osmolalities is therefore highly recommended since the deviation to the theoretical value might be even higher. Unless there are no limit values of osmolality for higher concentrated glucose solutions, the measurements can be used as a good example to show that the comportment of osmolality is not linear.
Additional Results
Fig. A1 Temperature-time-curve
Additional Materials and Methods
Tab. A1 Method parameters
Tab. A2 System configuration & data

References
- http://flexikon.doccheck.com/de/Ringer-L%C3%B6sung
- https://www.gesundheit.gv.at/labor/laborwerte/
- https://www.serag-wiessner.de
Application details


