Science with Passion
Application No.: VBS0064
Version 1 06/2017
Comparison of IgG purification with two different protein A media
Michaela Schulze, Kate Monks; applications@knauer.net
KNAUER Wissenschaftliche Geräte GmbH, Hegauer Weg 38, 14163 Berlin
Summary
The purification of antibodies is generally performed with protein A column materials. Two prepacked protein A columns from two different suppliers were compared in their ability to purify immunoglobulin antibodies (IgG) from human plasma. Capacity and yield were compared and revealed no significant differences in the performances of the two investigated columns.
Introduction
Today, the most widely used affinity chromatography purification procedure in industry is the capture of antibodies using protein A ligand. Affinity chromatography is used to isolate and enrich proteins or nucleic acids from complicated mixtures like human plasma. The principle is based on biospecific interactions between two reaction partners. The column matrix contains a covalently bound ligand to which the substance of interest specifically binds. A certain buffer is needed to elute the antibodies bound to the matrix. This type of chromatography is very efficient and delivers a highly clean protein. An AZURA® Bio purification system was used for comparison of two affinity materials. Here, an automated method to purify IgG from human plasma was used. Two protein A media were compared. The capacity of the columns to bind IgG was determined for both materials.
Results
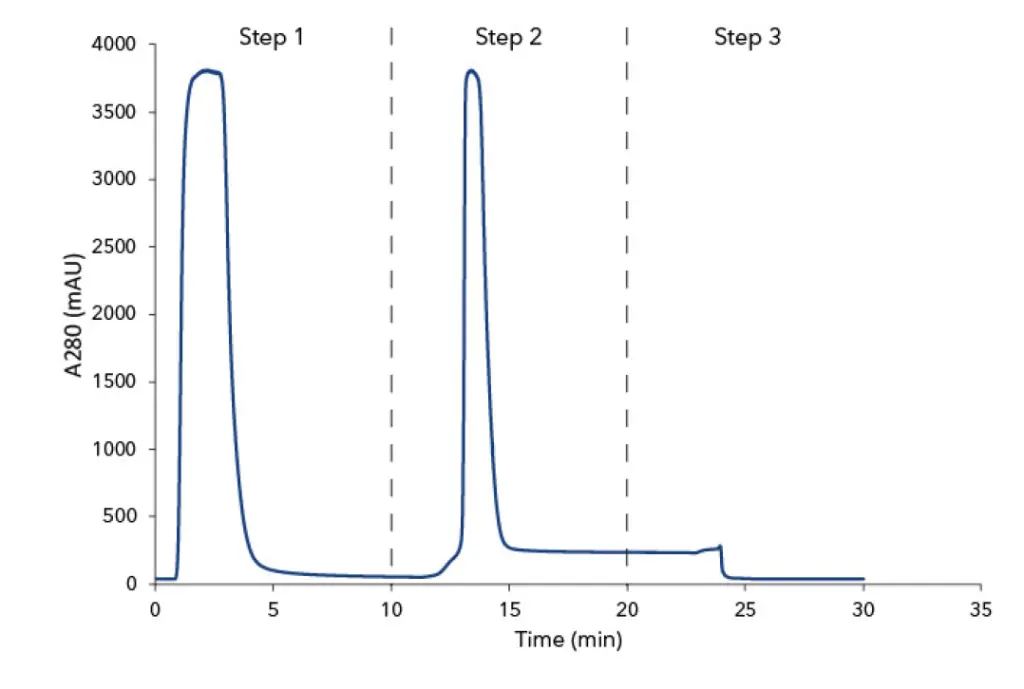
The IgG were purified from 500 µL human plasma by affinity chromatography using protein A columns. The eluted antibodies were then automatically collected with a fraction collector. The chromatogram of the whole purification process is divided in three steps (Fig 1 A&B). During the first step, the protein A column was equilibrated with buffer A followed by feed injection. The proteins that did not bind to the protein A columns went to waste and were visible as large flow through peak in step one. Thereafter, the column was washed with buffer A until no further peaks were detected. During step two, the antibodies were eluted from the column with buffer B and collected in 1 mL fractions. In the last step, the protein A columns were equilibrated with the buffer A in preparation for the next sample injection. Three runs were performed for each column material. The amount of purified protein was with 6 mg IgG originating from 500 µL human plasma are similar (Tab 1). Moreover, a SDS PAGE analysis was performed with the IgG samples to check the purity of the individual fractions. The protein bands of IgG heavy chains (HC) and IgG light chains (LC) are visible at 48 kDa and 25 kDa in the SDS-PAGE (Fig A1, suppl. material).
(A)
(B)
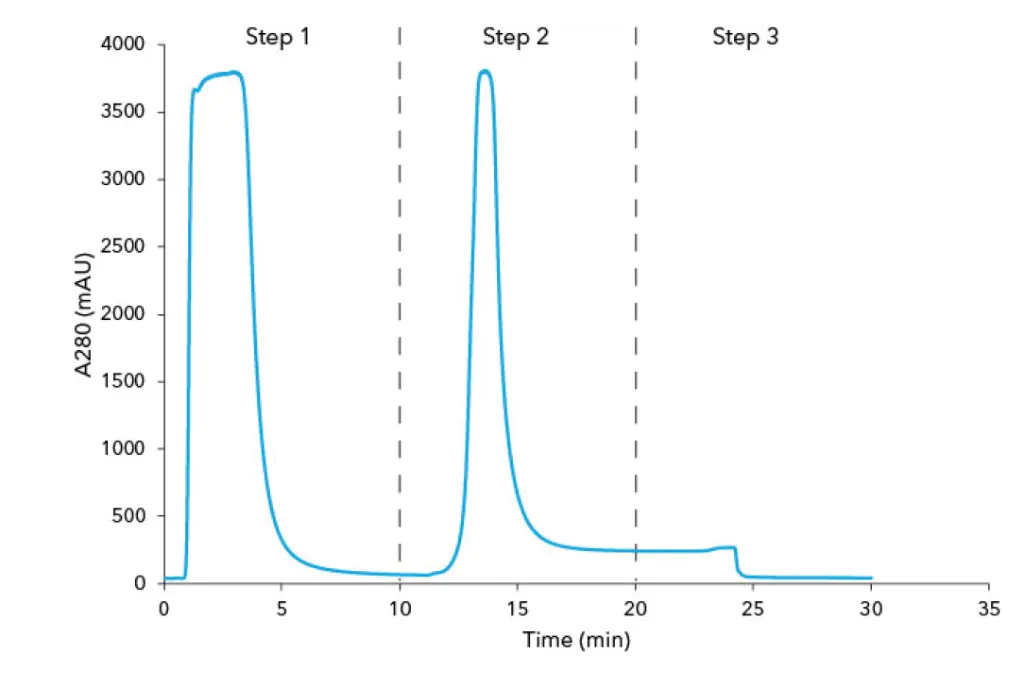
Chromatogram of antibody purification with Protein A HP column from vendor x (A) and Sepapure Protein A FF (B) columns; Step 1: injection peak of human plasma and column washing; Step 2: elution peak of IgG from protein A column with buffer B; Step 3: column equilibration with buffer A
Total and fraction concentrations of IgG purified with Sepapure Protein A FF or Protein A HP column from vendor x for three runs
Materials and Methods
In this application an AZURA Bio purification system was used. It consisted of an AZURA P 6.1L LPG metal-free pump, AZURA ASM 2.1L assistant module with feed pump and two 6 port/3 channel injection valves, an AZURA DAD 2.1L diode array detector with 10 mm, 10 µL flow cell cartridge; AZURA CM 2.1 conductivity monitor and a fraction collector.
The two 5 mL protein A columns were equilibrated with 20 mL buffer A (20 mM phosphate Buffer pH 7.0) at 2 mL/min. Then 500 µL human plasma were injected and the column washed with 20 mL buffer A at 2 mL/min.
The antibodies were then eluted with 20 mL elution buffer B (0.1 M Glycin-HCL, pH 2.7) and collected with the fraction collector. The concentrations of the IgG fractions from each individual run were determined with a NANODROP 2000 Tab 1). The UV signal was measured at 280 nm and conductivity signal was recorded.
Conclusion
Human immunoglobulin antibodies (IgG) were successfully purified with both protein A materials via affinity chromatography with the AZURA Bio purification system. An average of 6 mg IgG was purified from 500 µL of human plasma with both columns. When compared via SDS PAGE, in both cases eluted proteins are identical in purity. All in all, it can be concluded that the purification is quantitatively and qualitatively identical for both tested column materials.
References
[1] Janeway CA Jr, Travers P, Walport M, et al.; Immunology: The Immune System in Health and Disease. 5th Edition, New York; Garland Science, 2001
Additional Materials and Methods
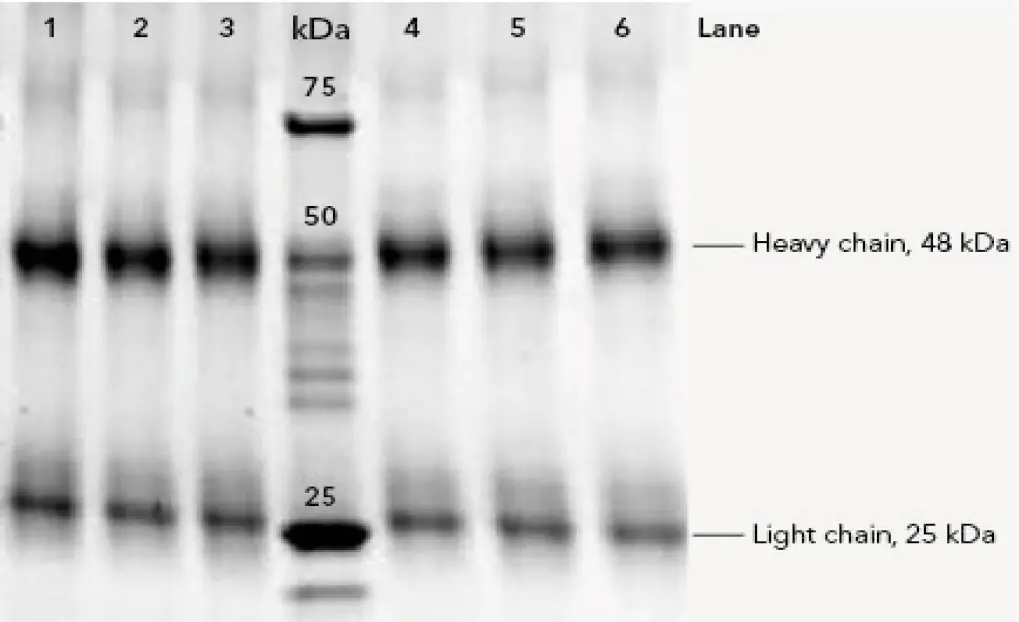
Fig. A1 SDS Page of the eluted IgG from human plasma; Lanes 1 – 3 IgG (4 µg) purified from three runs with the Sepapure Protein A FF column; Marker (10 µL) in kDa; Lanes 4-6 IgG (4 µg) puri-fied from three runs with the vendor x column
Tab. A1 Method parameters
Tab. A2 System configuration

Related KNAUER Applications
VBS0063 – Automated two – step purification of mouse antibody IgG1 with AZURA Bio purification system
VBS0067 – Automated two-step purification of 6xHis-tagged GFP with AZURA Bio purification system
VBS0068 – Fast and robust purification of antibodies from human serum with a new monolithic protein A column
VBS0066 – Fast and sensitive size exclusion chromatography of IgG antibody
VBS0069 – Purification of Sulfhydryl Oxidase
Anwendungsdetails