Overview of HPLC Detection Techniques – The Big Picture
High-performance liquid chromatography (HPLC) is one of the most widely used tools in analytical chemistry, making waves across diverse fields like pharmaceuticals, bioprocessing, environmental analysis, and polymer characterization. While the chromatographic separation is of course essential, it is only half of the story. To get meaning out of a separation, we need a detector that can convert the eluting analyte into measurable data.
Kicking off our HPLC detector blog series, we’ll explore the key detection principles and show you how to pick the perfect detector for your application.
Why Detection Matters
Put simply, the detector is the “eye” of the HPLC system. It keeps an eye on the eluate as each compound exits the column and translates their presence into electronic signals, that typically appear as peaks in a chromatogram.
When choosing a detector, several factors come into play:
- Sensitivity: How small a concentration can be reliably quantified. The higher the sensitivity, the smaller the amount of substance you can accurately quantify.
- Selectivity: Whether only certain analytes are detected or everything is.
- Linearity and Dynamic Range: How accurate results remain at low and high concentrations.
- Compatibility: How well it works with solvents, gradients, temperature, etc.
In practice, there’s no one-size-fits-all solution. The “best detector” depends on a combination of factors: the chemical nature of your analyte, the specific requirements of your application, and, of course, your budget.
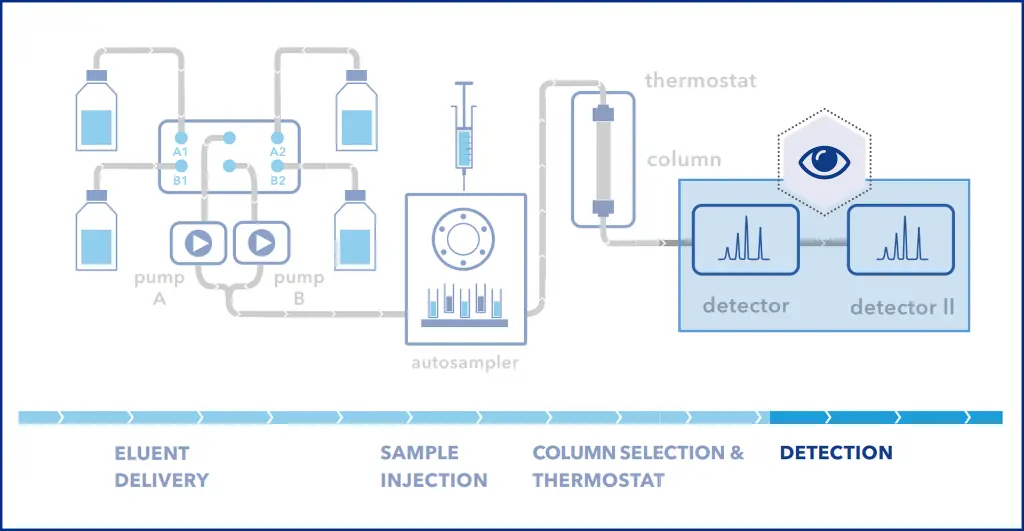
Figure 1: Typical HPLC system configuration with two detectors. (Graphic by KNAUER)
Major Detection Principles in HPLC
There are dozens of detector designs out there, but they typically can be put into five main categories:
- Optical detectors
- Mass-based detectors
- Electrochemical detectors
- Bulk property detectors
- Specialized or application-specific detectors
The techniques behind these detector types can be put into three general categories:
- Sample specific
- Bulk property
- Hyphenated techniques
Sample-specific detectors respond to particular characteristics of a sample that are unique to a specific sample or, at least, are not common among all analytes. In contrast, bulk property detectors measure a property that is shared by all compounds. They detect changes in this property by comparing the mobile phase containing the sample to that without the sample. Therefore, bulk property detectors can be considered universal. Hyphenated techniques basically refer to the combination of an independent analytical instrument with the HPLC system for enhanced detection capabilities.
Over the course of this blog series, we'll dive into one type of detector per post, talking about how it works, where it works best, and its limitations.
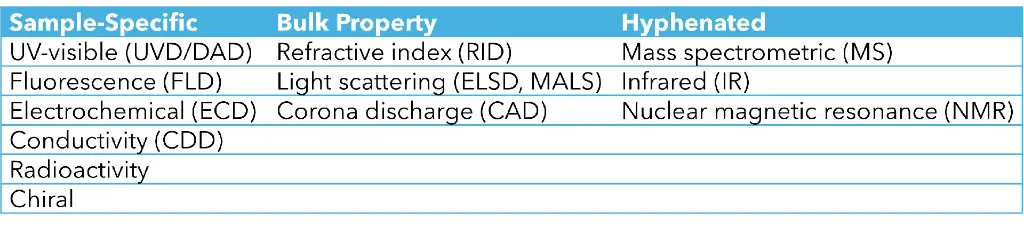
Table 1: Common HPLC Detectors. (Graphic by KNAUER)
1. Optical Detectors
Optical detectors measure how light interacts with the analyte. These are the most commonly used detectors in routine HPLC and include the following types:
- UV/VIS Absorption Detectors (UVD/DAD/PDA)
- Fluorescence Detectors (FLD)
- Optical Rotation or Circular Dichroism Detectors (for use in chiral applications)
- Light-Scattering Detectors (ELSD, CAD, MALS)
Optical methods are popular because they are sensitive, well-understood, and can be selective. For example:
- UV detects compounds with chromophores.
- FLD detects naturally fluorescent species or derivatized analytes.
- ELSD and CAD detect analytes without needing UV absorbance by measuring non-volatile analyte particles formed after evaporation of the volatile mobile phase.
You'll find these detectors are used a lot in pharmaceuticals, environmental analysis, polymers, and food science.
2. Mass Spectrometry Detectors
Mass spectrometry (MS) is pretty much the gold standard for detection in many fields, and for good reason! It provides:
- Very high sensitivity
- Ultra-high selectivity
- Structural information (mass and fragmentation patterns)
MS isn't a universal detector in the strict sense, it only detects compounds that can be ionized in the MS source, but its versatility, power for trace analysis, and ability to support both qualitative and quantitative work make it indispensable.
3. Electrochemical Detectors
Some compounds can undergo oxidation or reduction reactions at electrodes. Electrochemical detectors exploit this to provide highly sensitive detection, down to the picogram or even femtogram level for suitable analytes. Electrochemical detection is widely used for neurotransmitters, catecholamines, vitamins and other redox-active pharmaceuticals. Although highly selective, these detectors require exceptionally clean mobile phases and careful optimization.
4. Bulk Property Detectors
These detectors don't measure analyte properties directly but instead monitor a change in a physical property of the mobile phase when the analyte elutes. Examples include:
- Refractive Index Detector (RID)
- Viscosity Detectors
- pH Detectors
Although these kinds of detectors can be universal and are very useful when analytes lack chromophores, they generally have lower sensitivity and may be less compatible with gradient conditions.
5. Specialized Detectors
Certain applications require specific or specialized detectors with high selectivity, such as:
- Radioactivity Detectors (e.g. for tracer studies and nuclear medicine)
- MALS (Multi-Angle Light Scattering) for determining absolute molecular weight
- Chiral Detectors for enantiomeric purity
While these instruments tend to be more application-specific, they can provide uniquely powerful insights.

Figure 2: Detector types used in HPLC. (Graphic by KNAUER)
Final Thoughts
Even the best separation tells us nothing if we can't see it, that's why detection is one of the most important aspects of HPLC. Understanding detector principles helps you select the right detector type to truly reveal your results.
In the next blog post, we’ll take a closer look at why there are so many detectors out there, and what the main performance criteria and specifications are. This will help us to assess their relevance to particular chromatographic separations and make a more fair comparison among the different detectors.
For further information on this topic, please contact our author: huhmann@knauer.net
Resources
L. R. Snyder, J. J. Kirkland, J. W. Dolan, Introduction to Modern Liquid Chromatography, 3rd Edition, John Wiley & Sons, Inc., Hoboken, New Jersey, 2010.
D. A. Skoog, F. J. Holler, S. R. Crouch, Principles of Instrumental Analysis, 7th Edition, Cengage Learning, Boston, 2018.
R. P. W. Scott, Liquid Chromatography Detectors. In J. Chromatogr. Library, Vol. 11, Elsevier Scientific Publishing Company, Amsterdam, 1977, pp. iii-ix, 1-248.
V. R. Meyer, Praxis der Hochleistungs-Flüssigchromatographie, 10., vollst. überarb. u. erw. Auflage, Wiley-VCH, Weinheim, 2009.
G. Aced, H. J. Möckel, Liquidchromatographie, Apparative, theoretische und methodische Grundlagen der HPLC, VCH Verlagsgesellschaft mbH, Weinheim, 1991.

