
Science with Passion
Application No.: VFD0173 Version 1 05/2019
Be(e) wary – determination of neonicotinoid insecticides in honey
Juliane Boettcher, Kate Monks; applications@knauer.net
KNAUER Wissenschaftliche Geräte GmbH, Hegauer Weg 38, 14163 Berlin

Photo: unsplash
Summary
Neonicotinoids are active substances used in plant protection products to control harmful insects. They are systemic pesticides, which means that they are taken up by the plant and transported through its leaves, flowers, roots, and stems, as well as pollen and nectar. Neonicotinoids affect the central nerve system of insects, leading to eventual paralysis and death [1]. Three honey samples from different sources were analysed for neonicotinoid content according to current regulating guidelines via a fast and simple HPLC method.
Introduction
Neonicotinoids are one of the most widely used classes of pesticides [2]. Five neonicotinoid insecticides are approved as active substances in the EU for the use in plant protection products, namely clothianidin, imidacloprid, thiamethoxam, acetamiprid, and thiacloprid [1]. They are closely monitored by the European Commission. Because of the potential risk for bees, the use of three of the substances (imidacloprid, clothianidin, thiamethoxam) was restricted in 2013 (see Regulation (EU) No 485/2013) [3]. In April 2018, the European Commission banned these three neonicotinoids for the outdoor use and only the permit for usage in permanent greenhouse remains [4]. For acetamiprid the EFSA established a low risk to bees. A ban or further restrictions of this substance are neither scientifically nor legally appropriate. The fifth neonicotinoid, thiacloprid, is a candidate for substitution based on its endocrine disrupting properties [3]. In this application clothianidin, thiamethoxam, imidacloprid, and acetamiprid in honey samples are determined referring to the maximum residue levels which are specified in Commission Reg. (EU) 2017/671 [5], Commission Reg. (EU) 491/2014 [6] and Commission Reg. (EU) 2017/626 [7]. Three different honey samples have been tested. One of the samples was the KNAUER honey, produced from a bee colony located in the KNAUER garden. The other ones were commercially available canola honey and fruit blossom honey.
Results
A reversed phase method was developed where the four neonicotinoids are baseline separated. The method was optimized regarding temperature and gradient slope using DryLab simulation software. A calibration in a range from 0.5 µg/mL to 10 µg/mL was prepared. Fig 1 shows the separation of a mixed standard at a concentration of 10 µg/mL. The calibration showed a good linearity and all correlation coefficients are calculated as R2 >0.9996. Based on the measurement at a concentration of 0.5 µg/mL the LOD and LOQ were calculated. The calculated values for the single compounds are summarized in Tab A1 (additional information). Sample preparation was carried out using a citrate-buffered QuEChERS extraction. The recovery rate including sample preparation was determined at three different levels: LOQ, 2 x LOQ, upper end of calibration. For the compounds following recovery rates were calculated (averaged values over all levels): clothianidin 87 %, thiamethoxam 91 %, imidacloprid 92 % and acetamiprid 95 %. Furthermore, three different honey samples were analyzed regarding neonicotinoids. Fig 2 to 4 show the chromatograms of the QuEChERS extracted and cleaned samples. In one of three samples residues of clothianidin were detected but they were in the range of limit of detection and hence far below the maximum residue level of 0.05 mg/kg for honey and other apiculture products [8].
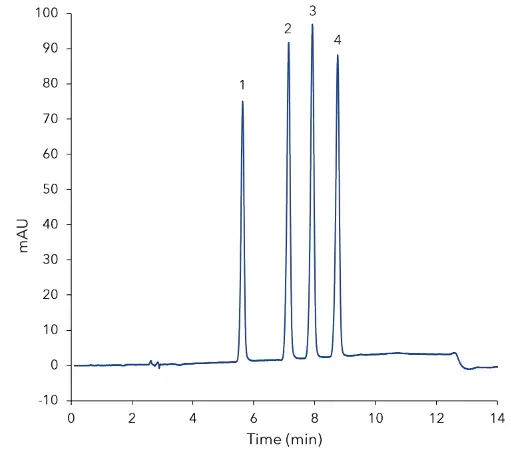
Fig. 1 Chromatogram of mixed standard at 10 µg/mL, 1) thiamethoxam, 2) clothianidin, 3) imidacloprid, 4) acetamiprid
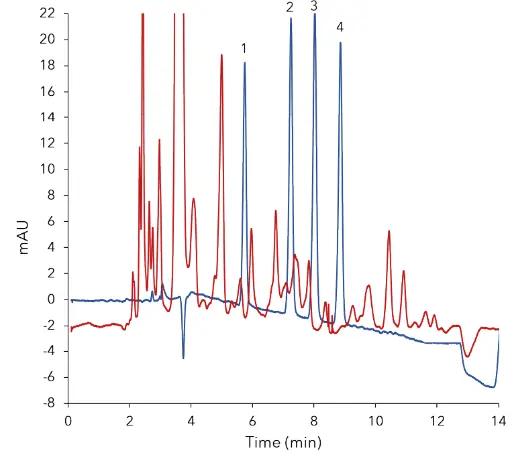
Fig. 2 Overlay of standard at 2.50 µg/mL (blue) and
cleaned sample of fruit blossom honey (red)
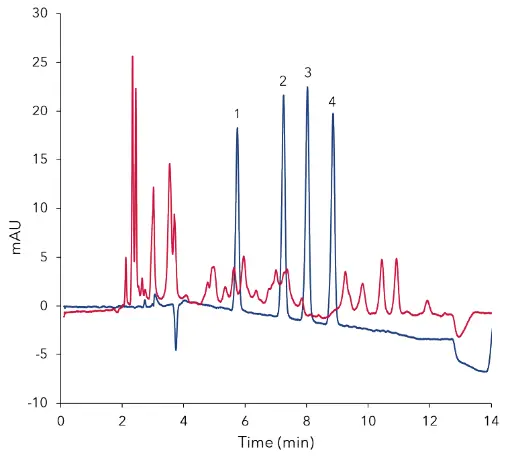
Fig. 3 Overlay of standard at 2.50 µg/mL (blue) and
cleaned sample of KNAUER honey (red)
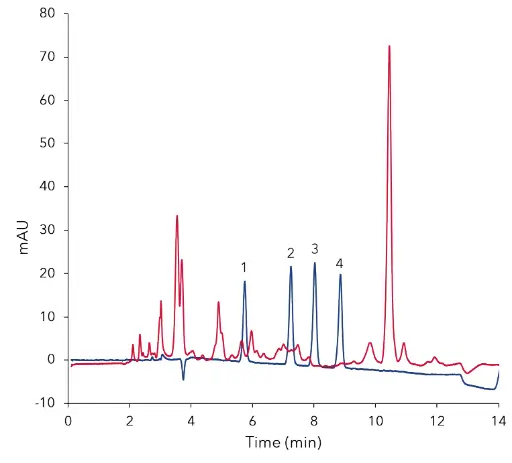
Fig. 4 Overlay of standard at 2.50 µg/mL (blue) and
cleaned sample of fruit blossom
Materials and Methods
The application was performed on an AZURA HPLC Plus System equipped with an AZURA P 6.1L HPG pump, AZURA CT 2.1 column thermostat, AZURA autosampler AS 6.1L and AZURA DAD 6.1L detector. The mobile phase was a composition of acetonitrile and water, both containing 0.1 % formic acid. The gradient method has a total run time of 15 minutes including equilibration. The flow rate was set to 1 mL/min. Temperature was set to 30 °C and detection was carried out at 260 nm with a data rate of 20 Hz. For the sample preparation BEKOlut QuEChERS Citrate-Kit-01 and PSA-Kit-02 were used. The QuEChERS extraction protocol is described in the additional results section. The used column in a dimension 250 x 4.6 mm ID was filled with Eurospher II 100 5 C18P silica.
Conclusion
Using QuEChERS extraction for sample preparation makes the handling of samples very easy and reduces time compared to e.g. solid phase extraction. Fortunately, neither the KNAUER honey nor the other tested samples were contaminated with neonicotinoids. Although banning neonicotinoids for the outside use, monitoring them is still mandatory. The developed method is suitable for quality control of honey or other apiculture products.
Additional Results
Tab. A1 LOD and LOQ values for single compounds
Additional Materials and Methods
Tab. A2 Method parameters
Tab. A3 Sample preparation
Tab. A4 System configuration & data
Related KNAUER Applications
VFD0161 - Determination of sugars in honey using HILIC separation and RI detection
VFD0169 - Determination of sugars in honey - comparison of refractive index and light scattering detection
VEV0012J - Determination of Carbamate Insecticides by HPLC with post-column derivatization
References
[2] Codling, G., et al. Concentrations of neonicotinoid insecticides in honey, pollen and honey bees (Apis mellifera L.) in central Saskatchewan, Canada, Chemosphere, Volume 144, 2016,Pages 2321-2328, ISSN 0045-6535,
https://doi.org/10.1016/j.chemosphere.2015.10.135.
[4] Official Journal of the European Union, L 132, 30 May 2018, https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=OJ:L:2018:132:TOC
[5] Commission Regulation (EU) 2017/671 of 7 April 2017 amending Annex II to Regulation (EC) No 396/2005,
http://data.europa.eu/eli/reg/2017/671/oj
[6] Commission Regulation (EU) No 491/2014 of 5 May 2014 amending Annexes II and III to Regulation (EC) No 396/2005, http://data.europa.eu/eli/reg/2014/491/oj
[7] Commission Regulation (EU) 2017/626 of 31 March 2017 amending Annexes II and III to Regulation (EC) No 396/2005, http://data.europa.eu/eli/reg/2017/626/oj
[8] Regulation (EC) No 396/2005 of the European Parliament and of the Council of 23 February 2005, http://data.europa.eu/eli/reg/2005/396/oj
Application details
|
Method |
HPLC |
|
Mode |
RP |
|
Substances |
Thiamethoxam, clothianidin, imidacloprid, acetamiprid |
|
CAS number |
153719-23-4, 210880-92-5, 138261-41-3, 135410-20-7 |
|
Version |
Application No.: VFD0173 | Version 1 05/2019 | ©KNAUER Wissenschaftliche Geräte GmbH |


