Science with Passion
Application No.: VFD0168 Version 1 03/2019
Oh so sweet – Quantification of steviol glycosides in stevia samples with RP-HPLC
**Johannes Menke, *Juliane Böttcher, *Kate Monks; applications@knauer.net
*KNAUER Wissenschaftliche Geräte GmbH, Hegauer Weg 38, 14163 Berlin
**Staatliche Technikerschule Berlin, Bochumer Str. 8B, 10555 Berlin

Summary
Steviol glycosides are the main sweetening compounds in Stevia rebaudiana and can be used as natural sugar substitutes. This method provides a fast determination of six steviol glycosides using the AZURA® HPLC Plus System. With a fast separation under 10 min and an optimized gradient, the developed method is suitable for a fast quality control of stevia products. Furthermore, the robustness for this method was validated using DryLab simulation software.
Introduction
Steviol glycosides are the main sweetening compounds in Stevia rebaudiana which have a far higher sweetening power than normal sucrose or glucose. The sweetness is estimated to be about 400 times higher. Stevia additives have been approved by the EU since 2011 as sweeteners for beverages and food. Good quality Stevia formulates usually do not have the bitter aftertaste as often other sweeteners do. Furthermore, due to their chemical structure they have no known harmful effect on overweight individuals or patients suffering from diabetes, making them an ideal substitute for household sugars. [1] [2]
A method as been developed able to quantify and qualify six of the 12 to 14 steviol glycosides that can be found in stevia plants. The substances determined are rebaudioside A, stevioside, rebaudioside C, dulcoside A, rebaudioside B, and steviolbioside. The method was optimized in terms of temperature and gradient slope using DryLab simulation software. Two different stevia samples were analyzed. Firstly, a sample obtained from dried Stevia rebaudiana leaves and secondly a sample obtained from a commonly sold Stevia sweetener.
Results
In Fig 1 the separation of the mixed standard of six steviol glycosides used for calibration at a level of 0.1 mg/mL for each compound is depicted. The resolution of the critical pair of peaks, namely, rebaudioside A and stevioside was very good and both components were baseline separated. All determined steviol glycosides were baseline separated from each other. The results of the sample measurement are displayed in Fig 2 and 3. The quantification of the compounds was achieved with high accuracy and precision. As shown in Tab A1 (additional results section), several of the calibrated compounds could be determined in both samples. According to the manufacturer of the stevia sweetener it should only contain rebaudioside A with a mass percentage of 3 %. The measurement shows that this is clearly not the case. Rebaudioside A was determined to be the main component with about 2.4 %, but also stevioside, rebaudioside C, and rebaudioside B were measured. The calculated amounts of steviol glycosides in the analyzed samples are summarized in Tab A1 (additional results section). Furthermore, 3D-data was recorded showing that no impurities eluted at the same time with the analytes. The recorded continuous spectrum from 200 to 700 nm shows only maxima at the wavelength of 210 nm ensuring clean separation. No trace of sample matrix can be seen.
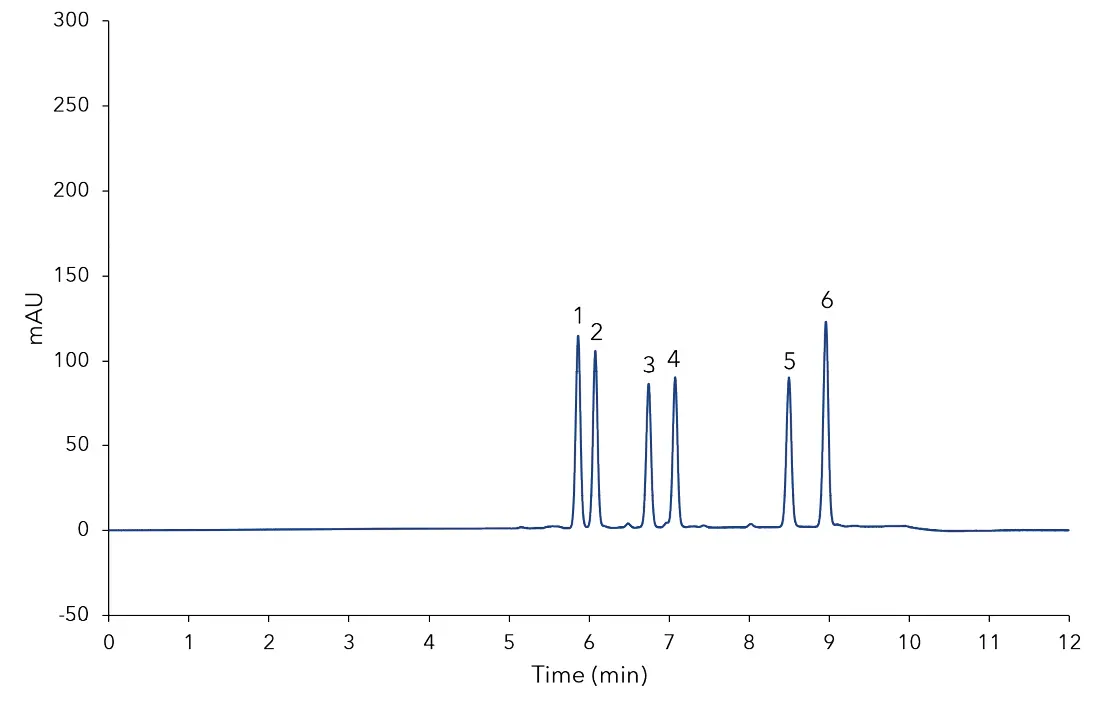
Fig. 1 Chromatogram of calibration standard at a concentration of 0.1 mg/m; 1) rebaudioside A, 2) stevioside, 3) rebaudioside C, 4) dulcoside A, 5) rebaudioside B, 6) steviolbioside
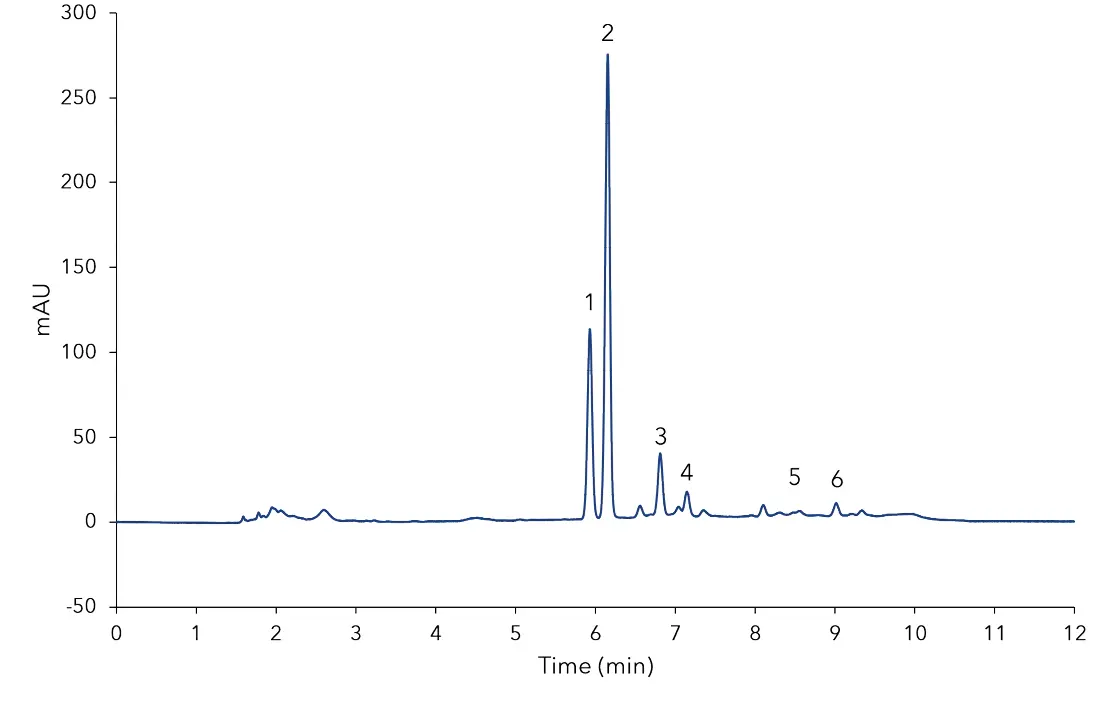
Fig. 2 Chromatogram of dried Stevia leaves sample after SPE purification;
1) rebaudioside A, 2) stevioside, 3) rebaudioside C, 4) dulcoside A, 5) rebaudioside B, 6) steviolbioside
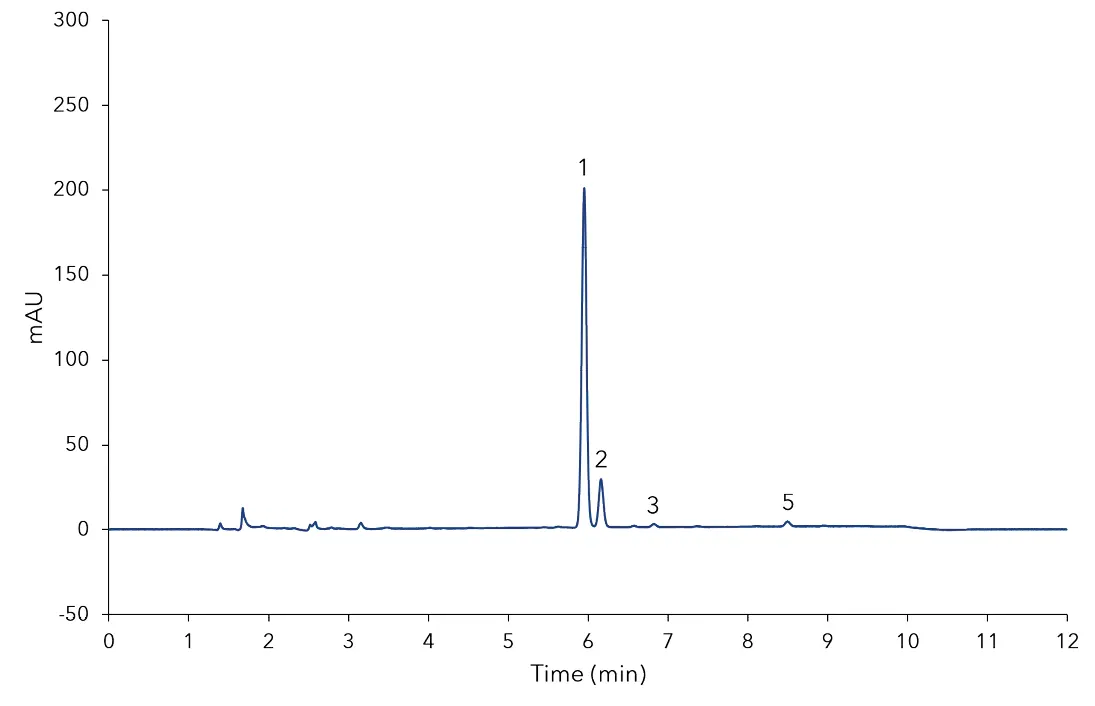
Fig. 3 Chromatogram of Stevia sweetener sample; 1) rebaudioside A, 2) stevioside, 3) rebaudioside C, 4) dulcoside A, 5) rebaudioside B, 6) steviolbioside
Materials and Methods
The determination of steviol gylcosides was performed on a KNAUER AZURA HPLC Plus System equipped with an autosampler AZURA AS 6.1L, a binary high-pressure gradient pump AZURA P 6.1L with 10 mL pump head, an AZURA CT 2.1L column thermostat, and a diode array detector AZURA DAD 2.1L. The eluent was a composition of A: water and B: acetonitrile. A step gradient at a flow rate of 1.2 mL/min was used with a total run time of 12 minutes including equilibration time. The column temperature was set to 40 °C. Detection was carried out at 210 nm and additionally the spectrum was recorded in a range from 200 nm to 700 nm. The column used here had the dimensions 250 x 4.6 mm ID with precolumn was filled with Eurospher II 100-5 C18 silica. Injection volume was 20 µL for samples and standard solutions.
Conclusion
The quantification of the compounds was achieved with high accuracy and precision again showing the robustness of this method. This method provides a fast and robust analysis for food samples containing steviol glycosides with a runtime of only 12 min per sample. Furthermore. with the optimized gradient and column temperature a baseline separation of the otherwise similarly eluting rebaudioside A and stevioside is achievable. The SPE sample preparation will diminish the matrix of plant extracts to a very low level preventing disturbances during sample analysis, which results in lower detection limits.
Additional Results
Tab. A1 Average amount. mass fraction and yield for dried stevia leaves and stevia sweetener samples
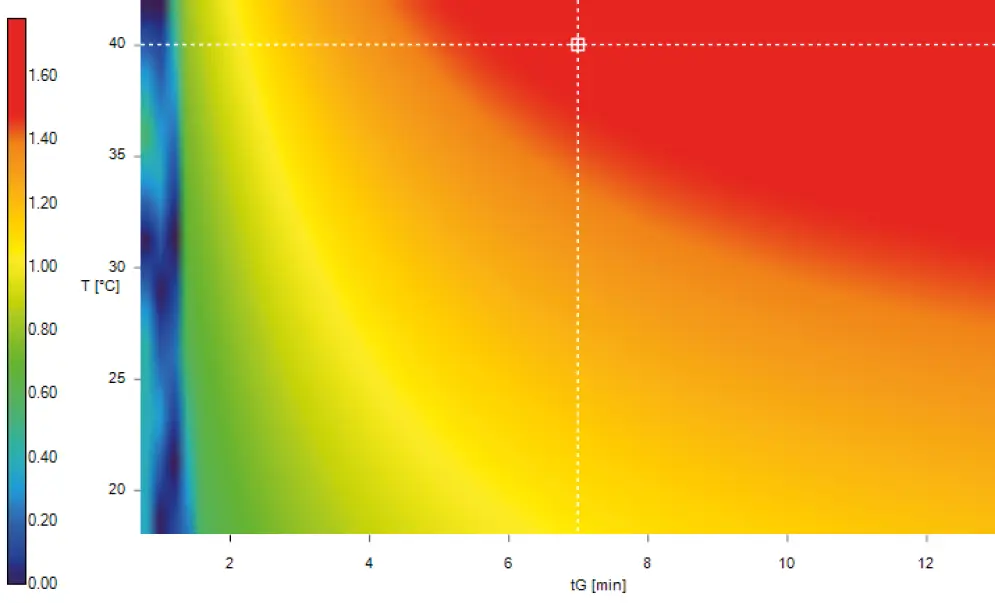
Fig. A1 Resolution map for optimized working point; red = good resolution, blue = poor resolution (Molnar Institute DryLab Version 4.3.1.1)
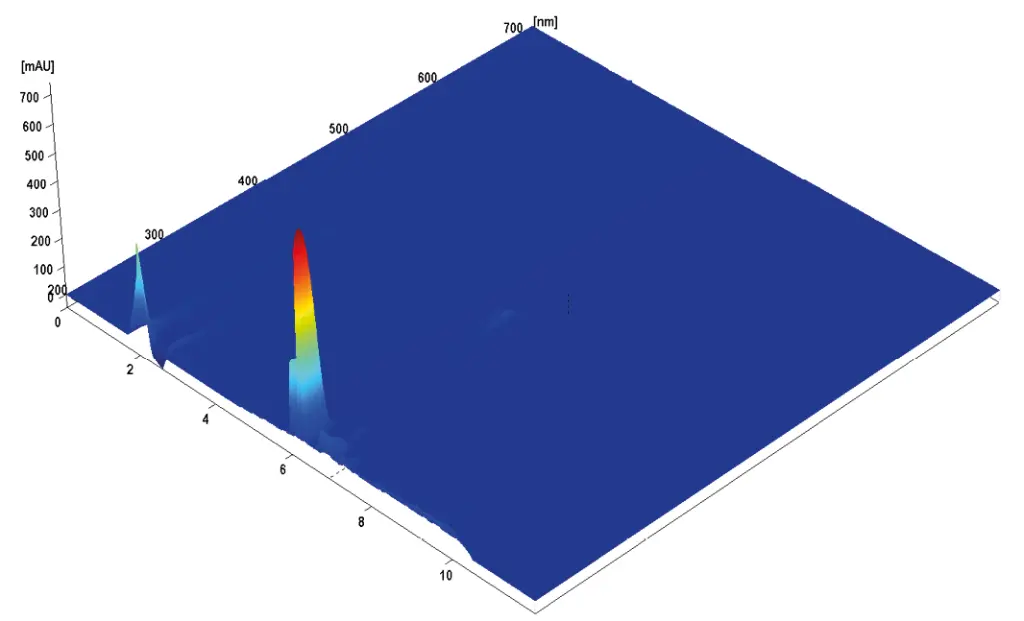
Fig. A2 Aquired PDA 3D Data for Dried Stevia Leaves sample (200–700 nm)
Additional Materials and Methods
Sample extraction:1 g of dried Stevia rebaudiana leaves were extracted with 25 mL distilled water at 60 °C and sonicated in an ultrasonic bath for 10 min following 30 min of extraction at 60 °C in a water bath. The mixture was centrifuged at 10,000 xg for 20 min at room temperature. The supernatant was transferred to a volumetric flask and the pellet was extracted two more times. The volume was adjusted with water to 100 mL. The extract was then filtered using 0.45 µm hydrophilic filter. 300 mg of common stevia sweetener powder were dissolved in 10 mL distilled water and then a lso filtered using a 0.45 µm hydrophilic filter. The solution was diluted 1:4 with water before analysis.
Solid phase extraction:The dried Stevia leaves extract was further purified using SPE-cartridges filled with 500 mg Eurospher II 100-20/45 µm C18 material. The cartridge volume (CV) was 3 mL. It was conditioned with 3 CV methanol and then washed with 1 CV water using a vacuum chamber. 2 mL of stevia extract were applied using gravitational force only. The cartridge was washed with 1 CV water and afterwards with 5 mL of 20:80 acetonitrile:water (v/v). For elution 2 mL acetonitrile:water 30:70 (v/v) was used.
Simulation: Parameters temperature (T) and gradient slope (tG) were optimized using DryLab simulation software. Corner stones of the simulation were four experiments: (1) T=40 °C, tG=30 min; (2) T=40 °C, tG=10 min; (3) T=20 °C, tG=30 min; (4) T=20 °C, tG=10 min. As sample calibration standard Level 4 was used.
Tab. A2 Method parameters
Tab. A3 System configuration & data
References
[1] Steviolglycoside in Süßwaren Entwicklung und Validierung einer Analysenmethode mittels HPLC-UV. Teresa Brandes. Marion Raters and Reinhard Matissek. DLR. 2013 literature\steviolglycoside-in-suesswaren-dlr.pdf
[2] Application Note – Fast online SPE purification of Stevia plant extracts. René Borstel. 2011 literature\vfd0093n_online_spe_of_steviol_glycosides.pdf
[3] Reversed-Phase HPLC Analysis of Steviol Glycosides Isolated from Stevia rebaudiana Bertoni. Venkata Sai Prakash Chaturvedula and Julian Zamora. Food and Nutrition Sciences. 2014. 5. 1711-1716 literature\Stevia_RP.pdf
Application details
|
Method |
HPLC |
|
Mode |
RP |
|
Substances |
Rebaudioside A, stevioside, rebaudioside B, rebaudioside C, dulcoside a, steviolbioside |
|
CAS number |
58543-16-1, 57817-89-7, 63550-99-2, 64432-06-0, 58543-17-2, 41093-60-1 |
|
Version |
Application No.: VFD0168 | Version 1 03/2019 | ©KNAUER Wissenschaftliche Geräte GmbH |