
Science with Passion
Application No.: VFD0165 Version 1 04/2019
Natural or artificial? – Determination of vanillin in vanilla products and associated marker substances
Juliane Böttcher, Kate Monks; applications@knauer.net
KNAUER Wissenschaftliche Geräte GmbH, Hegauer Weg 38, 14163 Berlin

Summary
Vanillin is one of the most popular flavouring agents used in various food products, beverages, as well as in the pharma and perfume industry. With a high demand for the supply of vanilla pods and the continuous increase in price, artificial vanilla flavouring agents of synthetic origin are nowadays highly requested [1]. With this application the components of different vanilla products can be analysed. This is useful i.a. for the first screening concerning the authenticity.
Introduction
The high demand for vanillin far exceeds the supply from all sources covered by vanilla orchids which are the only source for the “real“ vanilla flavour called “Bourbon vanilla”. The high price of natural vanillin, compared with that of synthetic vanillin, and the poor availability are the reasons to produce vanillin via chemical synthesis since the 1870s. These processes use coniferin, guaiacol, or eugenol as a precursor [2]. Biotechnological processes like fermentation that use ferulic acid and rice bran as precursors of vanillin are relatively new. Biotechnologically produced vanillin is much more cost intensive than chemically synthesized vanillin. However, but the biotechnically produced products are allowed to use the designation “natural vanilla flavour”. Chemically synthesized flavours must use the name “vanilla flavour”. Some substances from the chemical or biotechnological manufacturing processes are unwanted in food products due to negative health effects. This makes an analytical control indispensable. These molecules as well as the precursors used in the chemical synthesis are appropriate markers for the differentiation between synthetic vanilla flavour and Bourbon vanilla extract. While an exact statement about the origin of vanilla flavour is only possible after complex analytical methods like isotopic analysis, a first statement about the origin of vanilla flavour is already possible by screening for marker substances using HPLC methods. Therefore, in this work ethanolic extracts of vanillin containing samples are analysed to find marker substances as an association for the origin of the flavour.
Here, 4-hydroxybenzoic acid, vanillic acid, and 4-hydroxybenzaldehyde were analysed in addition to vanillin as typically occurring substances in Bourbon vanilla extract. Furthermore, guaiacol, coumarin, and eugenol were analysed as markers for synthetic vanilla flavour and unwanted precursors [3].
Results
A mixed standard of the seven compounds was used for calibration. The separation of the standard was achieved in under 4 minutes. All calibration curves showed a good linearity with R2 > 0.9999. The detailed concentrations for each level are summarized in Tab. 1.
Fig. 1 shows exemplarily the separation of the mixed standard at level 3. Four different samples were extracted and analysed: vanilla bean, bourbon vanilla sugar, vanillin sugar, and vanilla baking aroma. All samples were extracted with ethanol. The detailed sample preparation is described in the additional results section (Tab. A2).
Fig. 2 shows the chromatogram of the extracted vanilla bean sample. The sample profile shows a high amount of vanillin and as expected the marker substances for Bourbon vanilla origin.
4-hydroxybenzoic acid, vanillic acid and 4-hydroxybenzaldehyde were also measured. The total amounts of vanilla compounds are summarized and calculated in Tab. A1 (additional result section). Tab. 1 shows the determined values for LOD (S/N=3) and LOQ (S/N=10) for this method. The chromatograms of the other analysed samples are also displayed in the additional section.
Tab. 1 Concentration of calibration levels and calculated LOD and LOQ
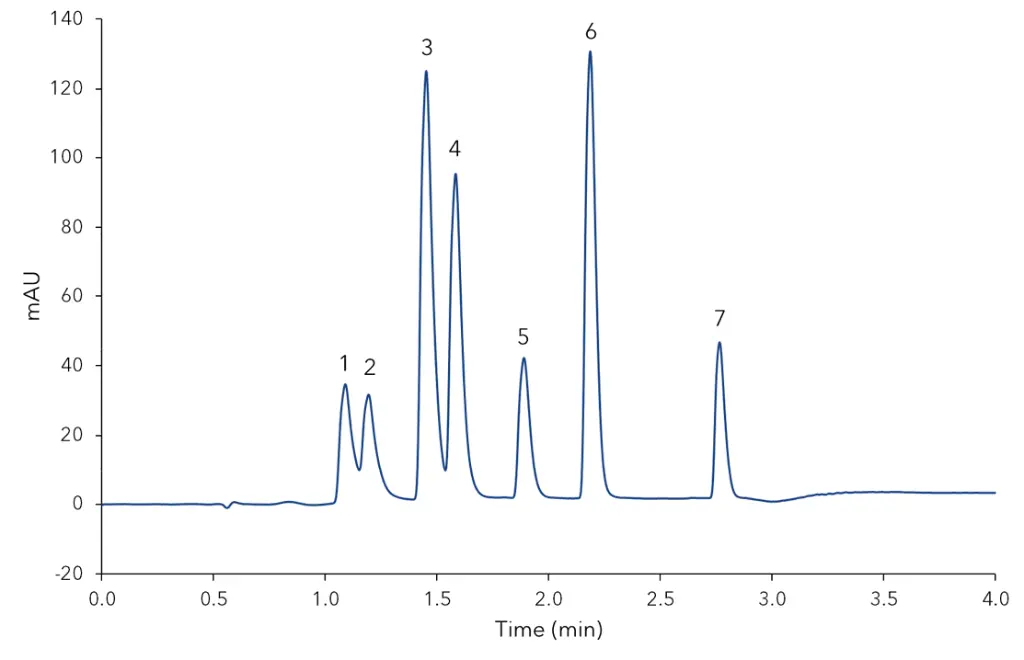
Fig. 1 Mixed standard at concentration Level 3, 1) 4-hydroxybenzoic acid, 2) vanillic acid, 3) 4-hydroxybenzaldehyde, 4) vanillin, 5) guaiacol, 6) coumarin, 7) eugenol
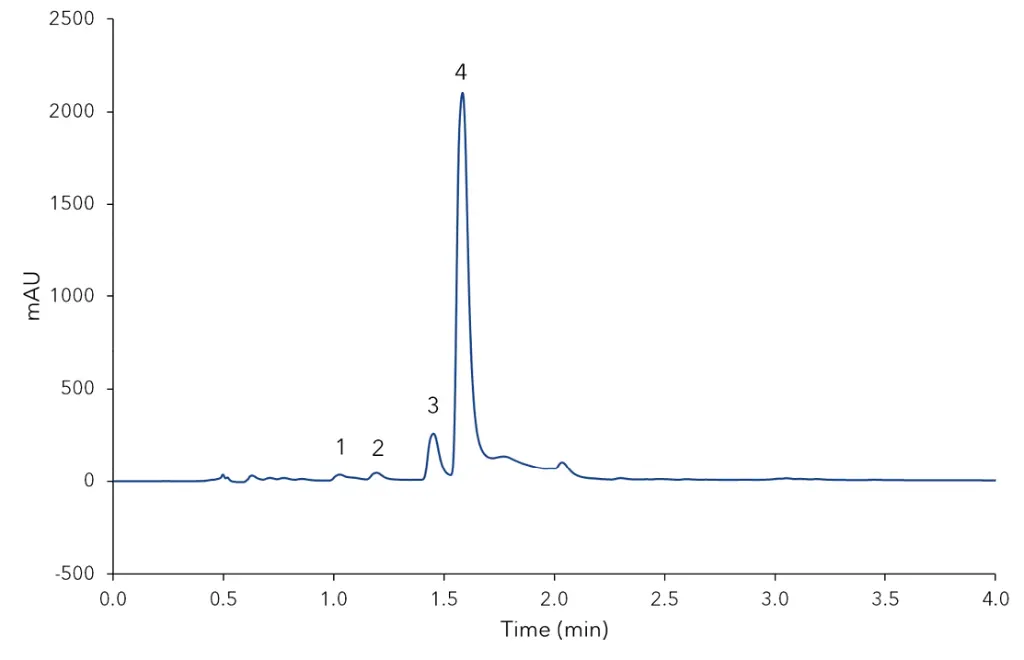
Fig. 2 Chromatogram of extracted vanilla bean, 1) 4-hydroxybenzoic acid, 2) vanillic acid, 3) 4-hydroxybenzaldehyde, 4) vanillin
Materials and Method
Here, the AZURA® UHPLC system was used which consisted of an AZURA P 6.1L HPG pump, an autosampler AZURA AS 6.1L, a column thermostat CT 2.1, and an AZURA MWD 2.1L. The flow was set to 0.5 mL/min at a temperature of 40 °C. 1µL of the samples and standards was injected. Detection took place at 280 nm. The mobile phase is a gradient composition of A: water with 0.05 % trifluoracetic acid and B: acetonitrile with 0.1 % trifluoracetic acid. The column was filled with Eurospher II 100-2 C18A silica in a dimension 100 x 2 mm ID.
Conclusion
According to the BLL guidelines for vanilla sugar and vanillin sugar from 2007 [4], vanilla sugar is a mixture of saccharose and crushed vanilla beans. Vanillin sugar, however, is a composition of saccharose and vanillin. Due to the calculated amounts in Tab. A1, the analysed Bourbon vanilla sugar contains vanillin as well as 4-hydroxybenzaldehyde, a marker for a natural vanilla flavour. The vanillin sugar on the other hand contains more vanillin and also a high amount of guaiacol which indicates its artificial/synthetic origin. The vanilla baking aroma contained the highest amount of vanillin but also residues of all other compounds. Since for the analysed aroma no declaration of composition is necessary, it could contain natural vanilla as well as synthetic aroma ingredients [5]. The shown UHPLC method allows a first and fast quality control of vanilla products regarding the marker substances for synthetic or natural based extracts. Besides the isotopic analysis there are characteristic numbers, also for HPLC analysis, that can be pulled to make a more sophisticated statement about the vanilla origin, but these were not considered in this application [6].
Additional Results
Tab. A1 Calculated amount of vanilla compounds (in mg/g)
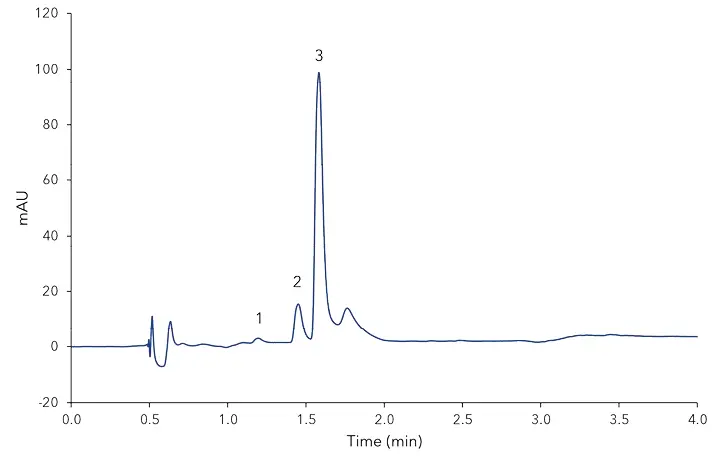
Fig. A1 Chromatogram of bourbon vanilla sugar, 1) vanillic acid, 2) 4-hydroxybenzaldehyde, 3) vanillin
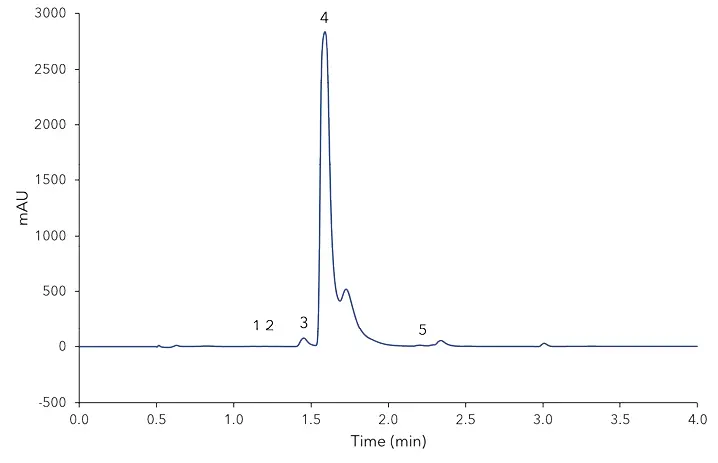
Fig. A2 Chromatogram of vanilla baking aroma, 1) 4-hydroxybenzoic acid, 2) vanillic acid, 3) 4-hydroxybenzaldehyde, 4) vanillin, 5) coumarin
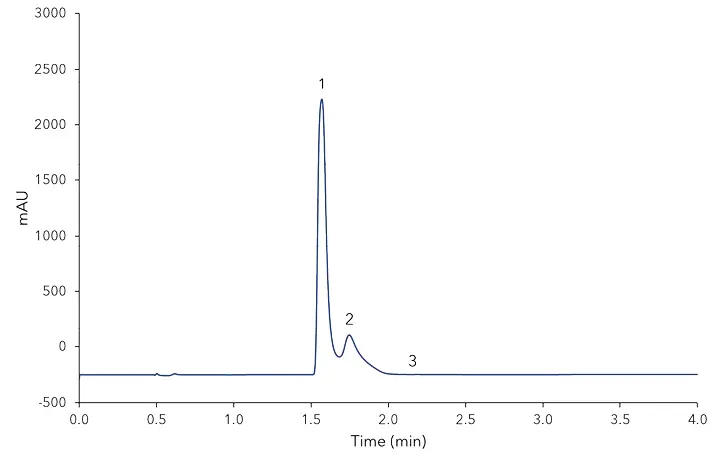
Fig. A3 Chromatogram of vanillin sugar, 1) vanillin, 2) guaiacol, 3) coumarin
Additional Materials and Methods
Tab. A2 Method parameters
Tab. A3 Sample preparation
Tab. A4 Pump parameters
Tab. A5 System configuration
References
[1] Krishna Veni et al, J Adv Sci Res, 2013, 4(1): 48-51: Analysis of Vanillin In Food Products By High Performance Thin Layer Chromatography
[2] Jagerdeo et al., Journal of AOAC International Vol. 83, No. 1, 2000 Liquid Chromatographic Determination of Vanillin and Related Aromatic Compounds
[3] Authenticity of vanilla and vanilla extracts, Elke Anklam, Joint Research Centre European Commission, Environment Institute Food & Drug Unit, 1993, EUR 15561 EN
[4] Richtlinie für Vanille-Zucker und Vanillin-Zucker (2007)
[5] Vanille und Vanillearomen, Vanille - die Königin der Gewürze
[6] Grundlagenpapier der Arbeitsgruppen „Aromastoffe“ und „Stabilisotopenanalytik“ in der Lebensmittelchemischen Gesellschaft zum Thema Herkunft und Authentizität von Vanillearomen
Related KNAUER Applications
VFD0136N – Comparison of compounds in Bourbon vanilla extract and vanilla flavor
VFD0073J – Determination of coumarin in cinnamon products
Application details
|
Method |
UHPLC |
|
Mode |
RP |
|
Substances |
4-hydroxybenzoic acid, vanillic acid, 4-hydroxybenzaldehyde, vanillin, guaiacol, coumarin, eugenol |
|
CAS number |
99-96-7, 121-34-6, 123-08-0, 121-33-5, 90-05-1, 121-32-1, 91-64-5, 97-53-0 |
|
Version |
Application No.: VFD0165 | Version 1 04/2019 | ©KNAUER Wissenschaftliche Geräte GmbH |



