Science with Passion
Application No.: VBS0069
Version 1 02/2018
Purification of Sulfhydryl Oxidase
Ulrike Krop, David Thiesing1, Tigran Kharatyan1, Jessica Thiesing-Paul,
Kate Monks; applications@knauer.net
KNAUER Wissenschaftliche Geräte GmbH, Hegauer Weg 38, 14163 Berlin;
1SternEnzym GmbH & Co. KG, Ahrensburg, Germany

Summary
Enzymes play a key role in food production. The use of recombinant enzymes for the food industry is a growing market. In this application we describe the purification of sulfhydryl oxidase (SOx) by affinity chromatography. Its stabilizing potential makes this enzyme an ideal candidate for food applications where proteins play a role, for example baking products or egg dishes. Additionally SOx is commercially rare and the broad application field provides huge potential for the food industry.
Introduction
With the recombinant expression of functional proteins through the development of modern biotechnology, enzymes have a special status in industry and research. Sulfhydryl oxidase (SOx) catalysis the formation of disulfide bonds within and between proteins which naturally plays a fundamental role for the folding of proteins during cell metabolism. In industrial food production, intermolecular crosslinks of proteins can have a stabilizing effect on products and could be used as a biological substitute for chemical stabilizers. [1] The future production and the commercial distribution is therefore of high interest.
Results
The recombinantly produced His-tagged SOx was purified with an immobilized metal ion chromatography (IMAC) resin. The chromatogram is shown in Fig. 1. Peak A represents the flow-through of unbound proteins. The His-tagged SOx was eluted with buffer B (Peak B) and collected via the fraction valve. The collected sample was analyzed by SDS-PAGE to check for impurities and evaluate the purity of the collected sample (Fig. 2). The recombinant His-tagged SOx has a molecular weight of 15 kDa. The supernatant shows a prominent 15 kDa band representing the expressed SOx. This band is not visible in the flow through fraction. Most of the His-tagged SOx bound to the IMAC column. Only minor contaminations are visible in the eluted protein fraction. A standard of SOx at a concentration of 1.13 g/L was prepared.
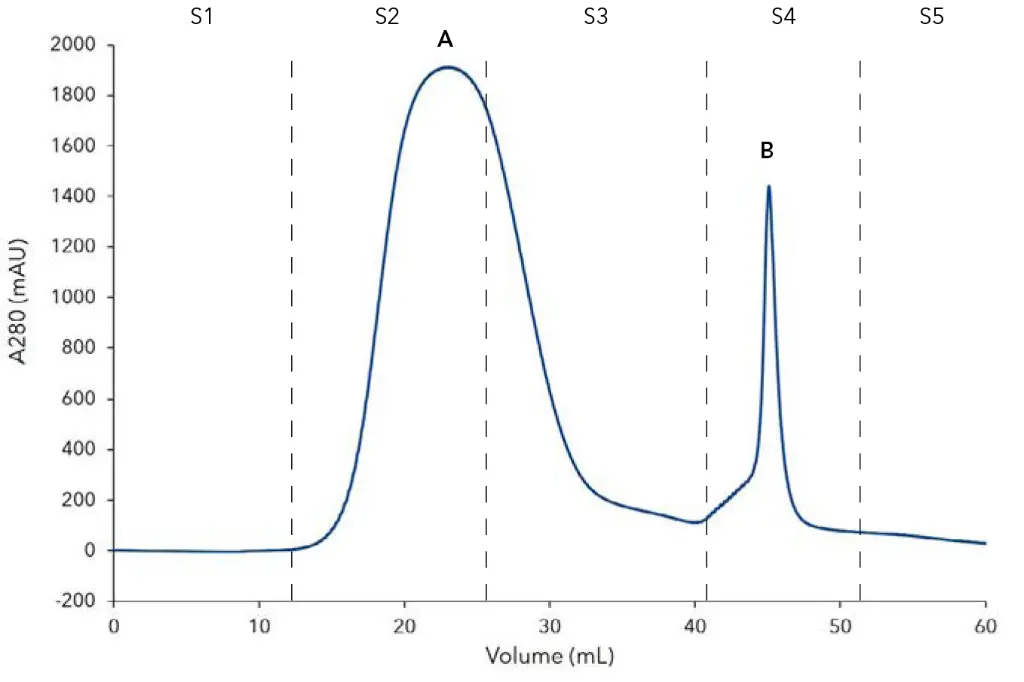
Fig. 1 Chromatogram of SOx affinity purification; A) flow through of unbound protein, B) elution peak of SOx; S1) column equilibration, S2) sample application, S3) column washing, S4 – elution of His-tagged Sox, S5 – re-equlibration
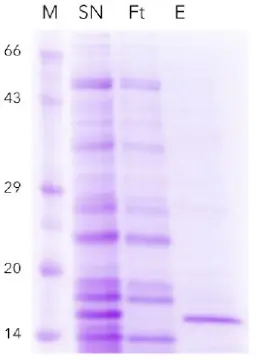
Fig. 2 SDS-PAGE of SOx purification,M) marker, SN) supernatant with overexpressed protein Ft) flow through, E) elutedprotein (purified SOx protein ~15 kDa)
Materials and Methods
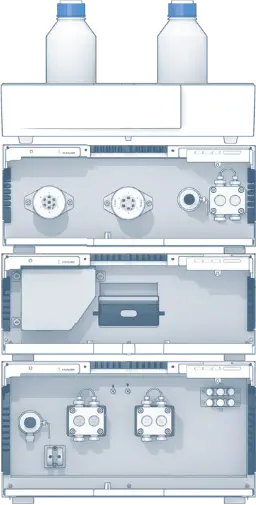
An AZURA® Bio purification system consisting of an AZURA P 6.1L HPG metal-free pump, AZURA ASM 2.1L assistant module 6 port/3 channel injection valve and a 6 port multi position fraction valve, an AZURA MWD 2.1L multi wavelength detector with semi-preparative biocompatible 3 mm, 2 µL flow cell cartridge was used. A Tricorn™ 10/200 column was filled with Chelating Sepharose™ Fast Flow to a column volume of 15 mL. The column was equilibrated with 50 mM NaOAc, pH 5.0 and loaded with nickel ions by applying 0.5 column volume 50 mM NaOAc, 100 mM NiSO4, pH 5.0. Unbound ions were washed out with 50 mM NaOAc, pH 5.0. Sulfhydryl-Oxidase was over expressed in Bacillus subtilis. After cultivation the fermentation broth was centrifuged for 30 min at 4300 x g for primary clarification purposes. The supernatant was 0.45 µm filtered, concentrated via ultrafiltration and subsequently used for the chromatographic purification. After applying the supernatant, the IMAC column was washed for 6 min at a flow rate of 3 mL/min with buffer A. Next, the target protein was eluted with 15 mL Buffer B and collected with the fraction valve. The column was re-equilibrated with buffer A. The UV signal was measured at 280 nm. The samples were analyzed for purity by SDS-PAGE.
Conclusion
Purification and concentration of SOx with the AZURA Bio purification system was successfully established. The recombinant His-tagged SOx was over-expressed in Bacillus subtilis and could be purified by IMAC from the fermentation supernatant. The availalability of pure enzyme enables tests for further characterization of the target enzyme as well as precise identification of the SOx’ potential in diverse food applications.
Additional Materials and Methods
Tab. A1 Method parameters
Tab. A2 System configuration
References
[1] Trivedi, M. V., Laurence, J. S., & Siahaan, T. J. (2009). The role of thiols and disulfides on protein stability. Current protein & peptide science, 10(6), 614–625.
Related KNAUER Applications
VBS0063 – Automated two step purification of mouse antibody IgG1 with AZURA Bio purification system
VBS0064 – Comparison of IgG purification by two different protein A media
VBS0067 – Automated two step purification of 6xHis-tagged GFP
VBS0066 – Fast and sensitive size exclusion chromatography of IgG antibody
Application details