Science with Passion
Application No.: VBS0066
Version 1 07/2017
Fast and sensitive size exclusion chromatography of IgG antibody
Lilit Avagyan, Michaela Schulze, Hagen Schlicke, Kate Monks;
applications@knauer.net
KNAUER Wissenschaftliche Geräte GmbH, Hegauer Weg 38, 14163 Berlin
Summary
Size exclusion chromatography (SEC) for analysis of antibodies like monoclonal immunoglobulin G (IgG) is a commonly used separation technique. AZURA® UHPLC system and TOSOH TSKgel UP-SW3000 silica based columns allow a faster and more sensitive determination of IgG compared to e.g. USP 129 application.
Introduction
The application field of monoclonal antibodies (mAb) goes from diagnostic kits up to active pharmaceutical ingredients for the treatment of autoimmune diseases and cancer. Since the commercialization of the first therapeutic antibody product in 1986, this class of biopharmaceutical products has grown significantly. Until 2020 about 70 new mAb products are predicted to be developed [1]. In the field of bio-chromatography, the research on separation of mAb increase accordingly. The SEC is the first-choice application for this purpose. However, the duration of the method is usually very long and the consumption of chemicals is high. Here, the increase of sensitivity and reduction of analysis time was reached by using silica based TOSOH TSKgel UP-SW3000 columns with 2 µm particle size and different column length. The analysis was performed on AZURA UHPLC system with AZURA DAD 6.1L diode array detector.
Results
Fig. 1 shows an overlay of two chromatograms, obtained by applying of IgG standard (diluted up to 1 mg/mL) on TSKgel UP-SW3000 columns with 300 mm (red) and 150 mm (blue) length. The retention time of the main peak representing IgG was recorded 6.61 and 3.59 min respectively (Tab. 1). The highest response value of 24.8 mAU/µg was reached with 150 mm length column. This is an increase by the factor of 1.55 and 7.51 compared to the 300 mm length column and to the certificated data respectively. The run time could be reduced from 30 (USP application) to 7 min (150 mm column). Compared to certificate data sheet and USP method (Tab. 1), the consumption of the sample was reduced up to four times.
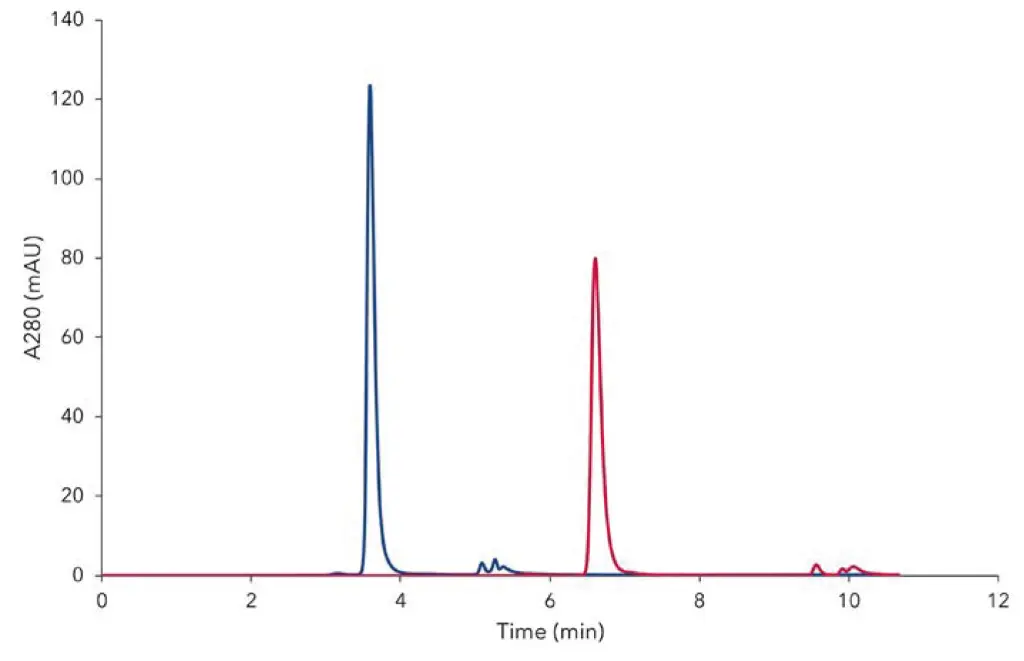
Fig. 1 Chromatograms overlay of IgG, obtained from measurement of diluted sample 1 mg/mL measured with 300 mm (red) and 150 mm (blue) columns, injection volume 5 µL
Tab. 1 Result table of IgG standard, measured via AZURA UHPLC system and TOSOH columns, compared to certificate data of standard.
Materials and Method
The AZURA UHPLC system consists of AZURA P 6.1L HPG pump, AZURA DAD 6.1L detector, LightGuide flow cell (10 mm, 2 µL), AZURA AS 6.1L autosampler, AZURA CT 2.1 thermostat and ClarityChrom software. As mobile phase a phosphate buffer with 0.14 M monobasic potassium phosphate, 0.06 M dibasic potassium phosphate and 0.25 M potassium chloride (pH 6.2) was used. The standard "monoclonal IgG system suitability" (USP catalog No. 1445550) was obtained by Sigma Aldrich. The stock solution (10 mg/mL) was diluted with mobile phase and used for analyses. The separation was applied on two TSKgel UP-SW3000 columns with 2 µm particle size and 4.6 × 300 mm and 4.6 × 150 mm column dimensions. The measurements were performed in isocratic mode with 0.4 mL/min flow rate. The injection volume was 5 µL. Determination took place at 280 nm, sampling rate of 20 Hz and time constant 0.05 sec. The column thermostat was adjusted to 25 °C. For characterization of sensitivity the response (R) of the signals was calculated according to the following equation, where H is the height of the peak (in mAU) and m is the mass of the sample (in µg):
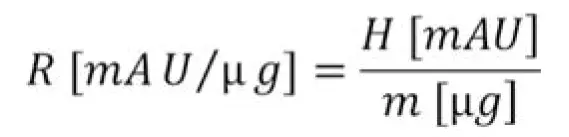
Conclusion
As the results show the application for determination of IgG could be significantly improved concerning run time and sensitivity by the using of AZURA UHPLC system and TOSOH TSKgel UP-SW3000 silica based columns.
Additional Results
Fig. A2 depicts a chromatogram of stock solution of IgG standard. 5 µL of the standard was injected on 300 mm TSKgel UP-SW3000 column. The obtained peak maximum value is 615.3 mAU (Fig. A2). Compared to certificate data sheet (Fig. A1) and USP method (Tab. 1), the consumption of the sample was reduced up to 4 times.
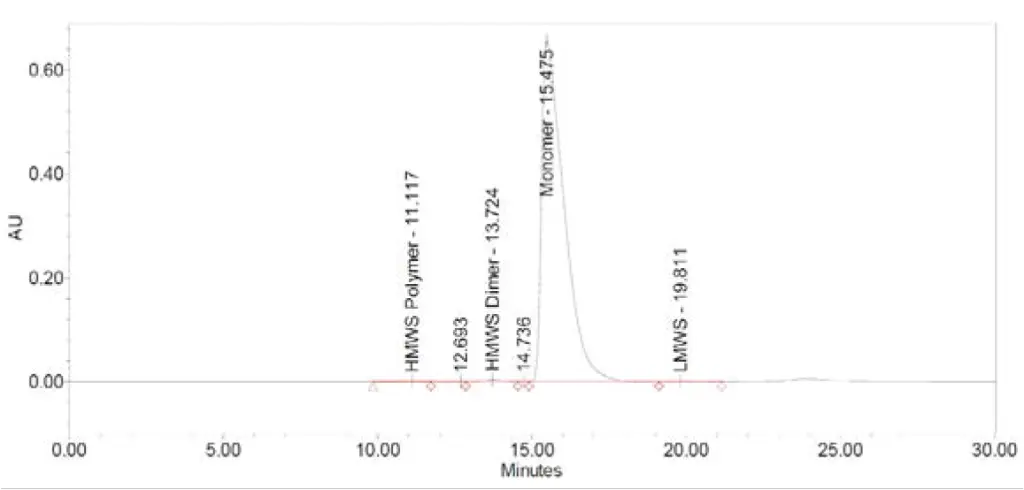
Fig. A1 Chromatogram of IgG, obtained from USP-standard certificate. Sample concentration 10 mg/mL, injection volume 20 µL
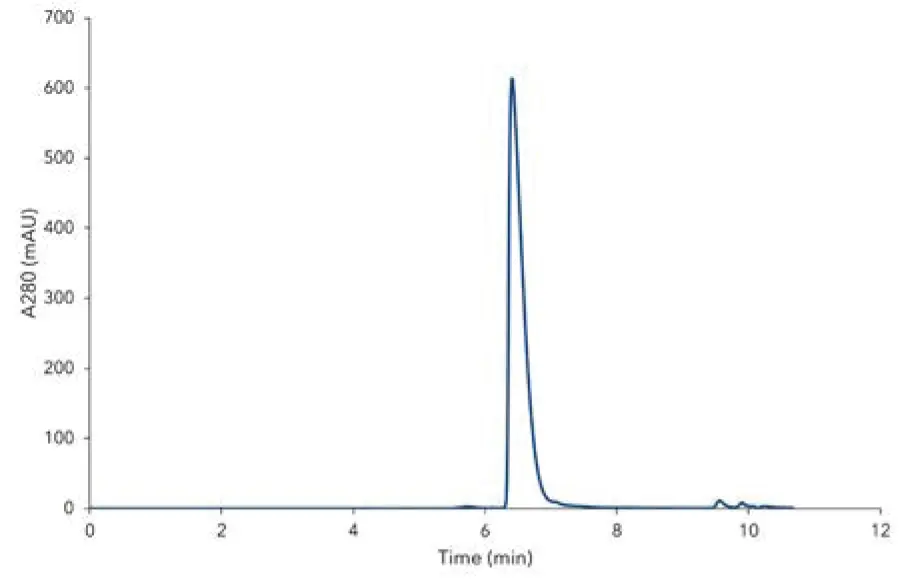
Fig. A2 Chromatogram of IgG, obtained from measurement of stock solution. Sample concentration 10 mg/mL, injection volume 5 µL
Additional Materials and Methods
Tab. A1 Method parameters
Tab. A2 System configuration
References
[1] Dawn M Ecker, Susan Dana Jones and Howard L Levine, Jones SD, Levine HL, The therapeutic monoclonal antibody market, MAbs. 2015 Jan-Feb; 7(1): 9–14.
Related KNAUER Applications
VBS0063 – Automated two-step purification of mouse antibody IgG1 with
AZURA Bio LC Lab system
VBS0064 – Comparison of IgG purification by two different protein A media
VBS0067 – Automated two-step purification of 6xHis-tagged GFP with
AZURA Bio LC
VBS0068 – Fast and robust purification of antibodies from human serum with a new monolithic protein A column
VBS0069 – Purification of Sulfhydryl Oxidase
Application details