
Science with Passion
Application No.: VBS0067
Version 1 11/2017
Automated two step purification of 6xHis-tagged GFP
Ulrike Maschke, Yannick Krauke, Kate Monks; applications@knauer.net
KNAUER Wissenschaftliche Geräte GmbH, Hegauer Weg 38, 14163 Berlin
Summary
Affinity chromatography by His-tag is one of the most widespread purification techniques for recombinant proteins. In most cases it requires an additional cleaning/polishing step. This application highlights the possibility of combining two subsequent chromatography protocols without manual interaction using the AZURA® Bio purification system.
Introduction
Affinity chromatography (AC) is one of the most efficient techniques to purify recombinant proteins. Mostly, AC is performed on crude samples like bacterial lysates containing the recombinant protein that is genetically engineered to be expressed with a tag that enables the specific capture of the recombinant protein. These highly efficient tags are used for affinity binding to specific affinity chromatography materials. A variety of tags is available among which the polyhistidine tag is the most widespread one. In this application, six histidine (6xHis) residues were attached to the green fluorescent protein (GFP). The histidine residues bind with very high affinity to the immobilized metal ions on the column (immobilized metal ion affinity chromatography (IMAC)). In many protocols, an additional step is recommended to reach higher purity or to change the buffer of the purified protein to a suitable storage buffer. Here, size exclusion chromatography was used as second step to exchange the buffer of the purified protein. Purification of recombinant proteins can be performed manually or by using a chromatography system combining two steps automatically to save time and effort.
Results
The chromatogram of the 6xHis-GFP purification shows the five phases of the two-step protocol (Fig. 1). After equilibration (Fig. 1, phase 1) the lysate was injected and the GFP bound to the Ni-NTA affinity column via the 6xHis-tag. All other non-binding proteins and impurities are in the large flow through peak (Fig. 1, phase 2, peak A). Subsequently, the column was washed until the baseline was stable (Fig. 1, phase 3). The eluted protein (Fig. 1, phase 4, peak B1) was collected in a sample loop and re-injected on the desalting column (Fig. 1, phase 5) to exchange the buffer from high imidazole concentrations to a buffer without imidazole. The purified protein (Fig. 1, peak B2) was collected by the fraction collector. Additionally to the unspecific photometrical detection of all proteins at 280 nm, GFP-signal was recorded at 395 nm (Fig. 2) with the multi-wavelength detector. Most of the 6xHis-tagged GFP bound to the column as only a small peak for GFP is visible in the flow through. The purification results were confirmed by SDS-PAGE (Fig. 3). The cell lysate (Fig. 3, lane 1) shows a prominent band representing the overexpressed 6xHis GFP. This band is cleared in the flow through (Fig. 3, lane 2), confirming that most of the tagged protein bound to the column. The eluted sample (Fig. 3, lane 3) shows the purified 27 kDa 6xHis-GFP with only minor contaminations.
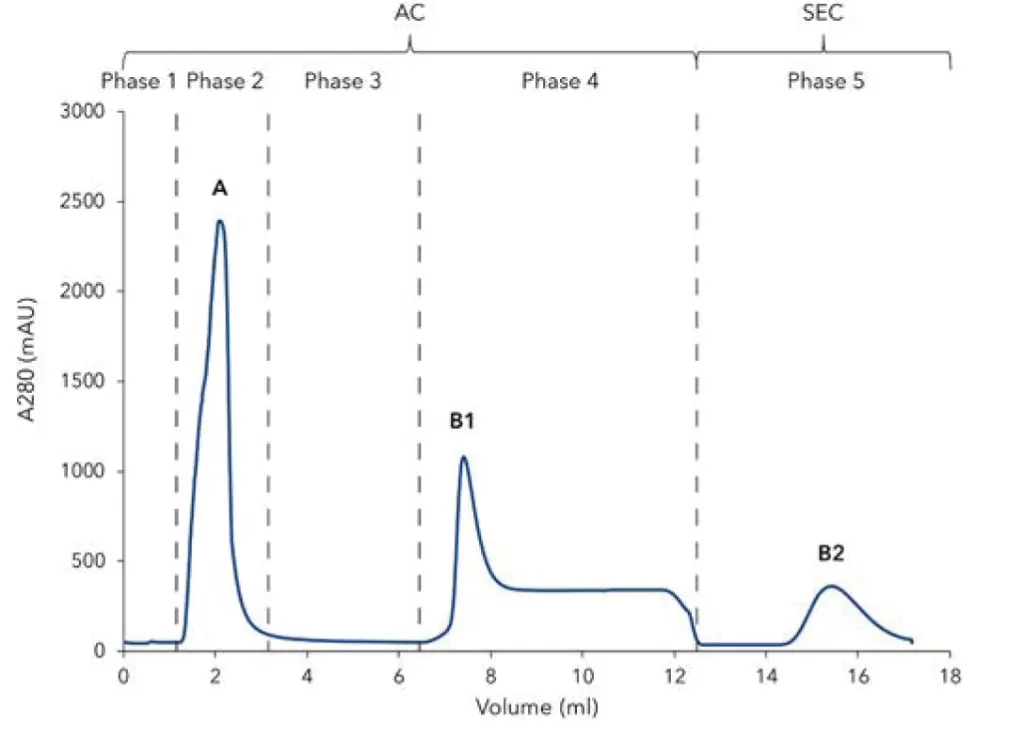
Fig. 1 Chromatogram of the two-step 6xHis-GFP purification; 280 nm UV signal. Step 1) Affinity chromatography (AC) / Ni-NTA column: 1) Column equilibration; 2) Sample injection; 3) Column washing; 4) Elution of 6xHis-GFP and parking in 1 mL sample loop. Step 2) Buffer exchange with desalting column: 5) Elution of 6xHis-GFP. A) flow through of unbound protein; B1) elution peak of 6xHis-GFP from Ni-NTA column; B2) elution peak of 6xHis-GFP.
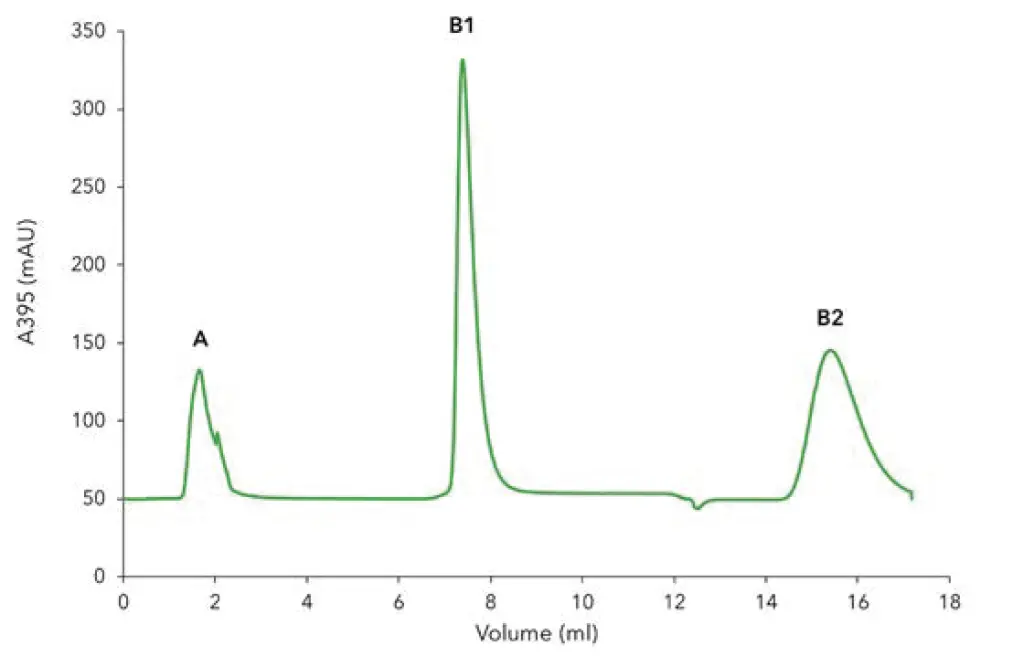
Fig. 2 Chromatogram of the two-step 6xHis-GFP, GFP detection with 395 nm UV signal. A) flow through of unbound protein; B1) elution peak of 6xHis-GFP from Ni-NTA column; B2) elution peak of 6xHis-GFP.
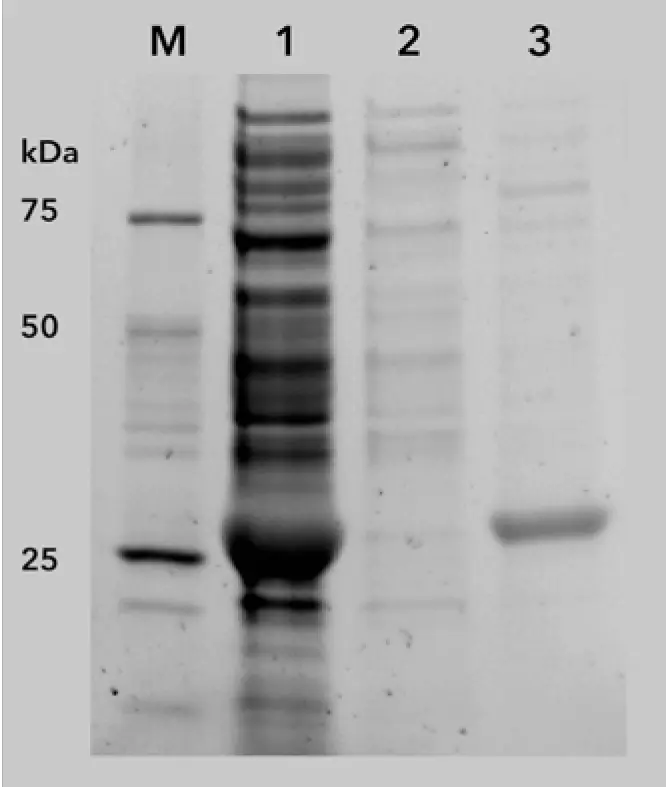
Fig. 3 SDS-PAGE of two-step 6xHis-GFP purification. M – marker; 1) lysate before purification; 2) flow through; 3) eluted 6xHis-GFP (27 kDa) after two-step purification.
Conclusion
6xHis-tagged GFP was purified by an automated two-step protocol combining an affinity chromatography method to capture 6xHis-tagged GFP with a subsequent buffer exchange step by size exclusion chromatography. This automatization requires no time consuming manual interaction. The method set up is an excellent example for a two-step protein purification and can be adapted to a variety of protein purification protocols. The benefit of a multi wavelength detector was shown measuring at two different wavelengths.
Materials and Methods
The AZURA two-step purification system with a multi-wavelength detector was used for this application. It consists of AZURA P 6.1L HPG, one autosampler AZURA ASM 2.1L with feed pump and two 6 port/3 channel injection valves; a second ASM 2.1L with two 6 port/3 channel injection valves; the MWD 2.1L multi-wavelength detector, a column switching valve, a conductivity monitor, and a fraction collector. The Sepapure FF Ni-NTA 1 mL column was equilibrated prior to the run with 15 mL load/wash buffer (PBS pH 7.5, 10 mM imidazole) at 1 mL/min. 100 µL lysate containing the 6xHis tagged GFP was loaded on to the column at a flow rate of 0.3 mL/min. The column was washed with 4 mL load/wash buffer at a flow rate of 1 mL/min. The load/wash buffer had a low amount of imidazole to reduce non-specific binding of impurities. 6xHis tagged GFP was eluted with 5 mL elution buffer (PBS, pH 7.5, 500 mM imidazole) and collected in a 1 mL sample loop. The eluted protein was re-injected on to a 5 mL desalting column to exchange the buffer from high imidazole concentrations from the elution buffer to the final desalting buffer without imidazole. 7 mL desalting buffer (PBS pH 7.4) was used for the gel filtration run at a flow rate of 1 mL/min. The protein was collected in a fraction collector. The UV signal at 280 nm and 395 nm, as well as the conductivity signal were recorded.
Additional Materials and Methods
Tab. A1 Method Parameters
Tab. A2 System Configuration
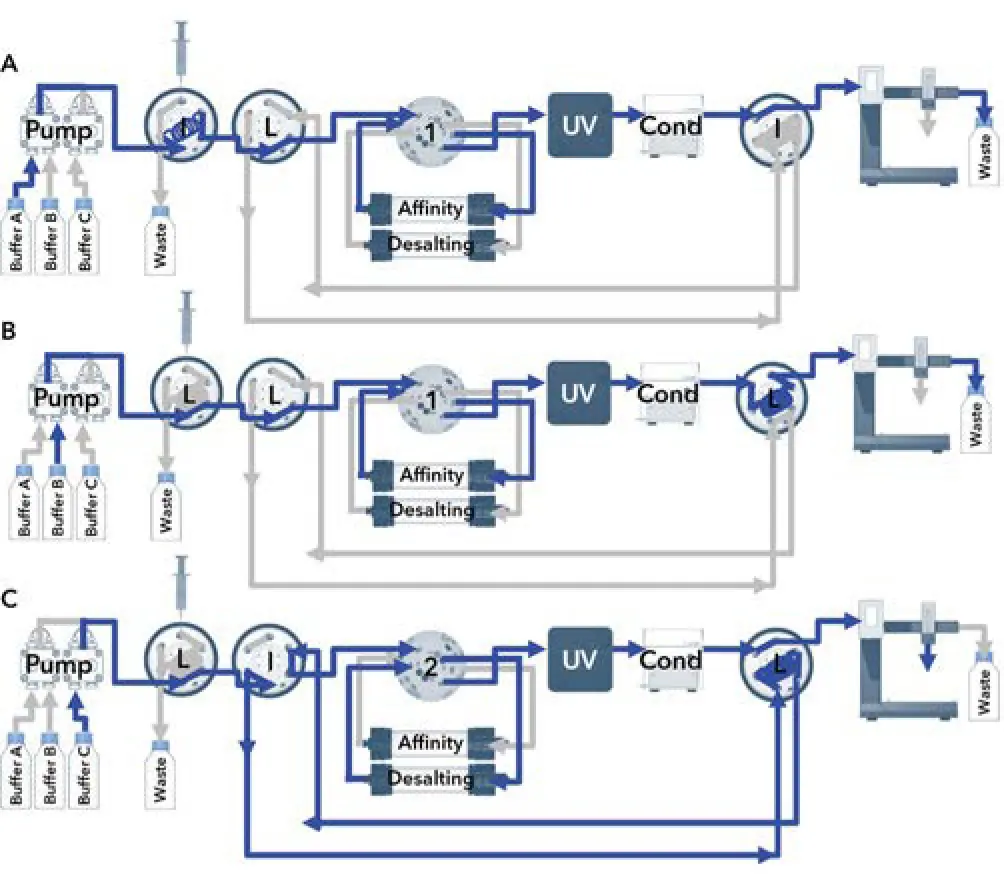
Fig. A1 Flowchart; A) sample injection, B) elution and peak parking, C) reinjection.
Related KNAUER Applications
VBS0063 – Automated two-step purification of mouse antibody IgG1 with AZURA Bio purification system
VBS0064 – Comparison of IgG purification by two different protein A media
VBS0066 – Fast and sensitive size exclusion chromatography of IgG antibody
VBS0068 – Fast and robust purification of antibodies from human serum with a new monolithic protein A column
VBS0069 – Purification of Sulfhydryl Oxidase
Anwendungsdetails



