Understanding Refractive Index (RI) Detection in HPLC – Seeing the Unseen
When people think of HPLC detectors, UV-Vis absorbance systems usually steal the spotlight, and for good reason. They’re sensitive, selective, and widely applicable. But what if your analyte doesn’t absorb in the UV or visible range?
That’s where the Refractive Index Detector (RID) comes in. Quiet, dependable, surprisingly versatile, and often considered the "universal detector" of HPLC, it can "see" compounds invisible to other detectors. If you’re analyzing compounds like sugars, polymers, lipids, or alcohols, you’ve likely used one or wondered whether you should.
Let’s take a closer look at how RI detection works, where it shines, and how to get the best performance from it.
💡How RI Detection Works
When light moves from one medium into another, it bends (or refracts) depending on the phase velocity and the angle of incidence. This behavior is described by Snell’s law, which shows that the degree of refraction is determined by the refractive index (RI) of the medium (see Figure 1).
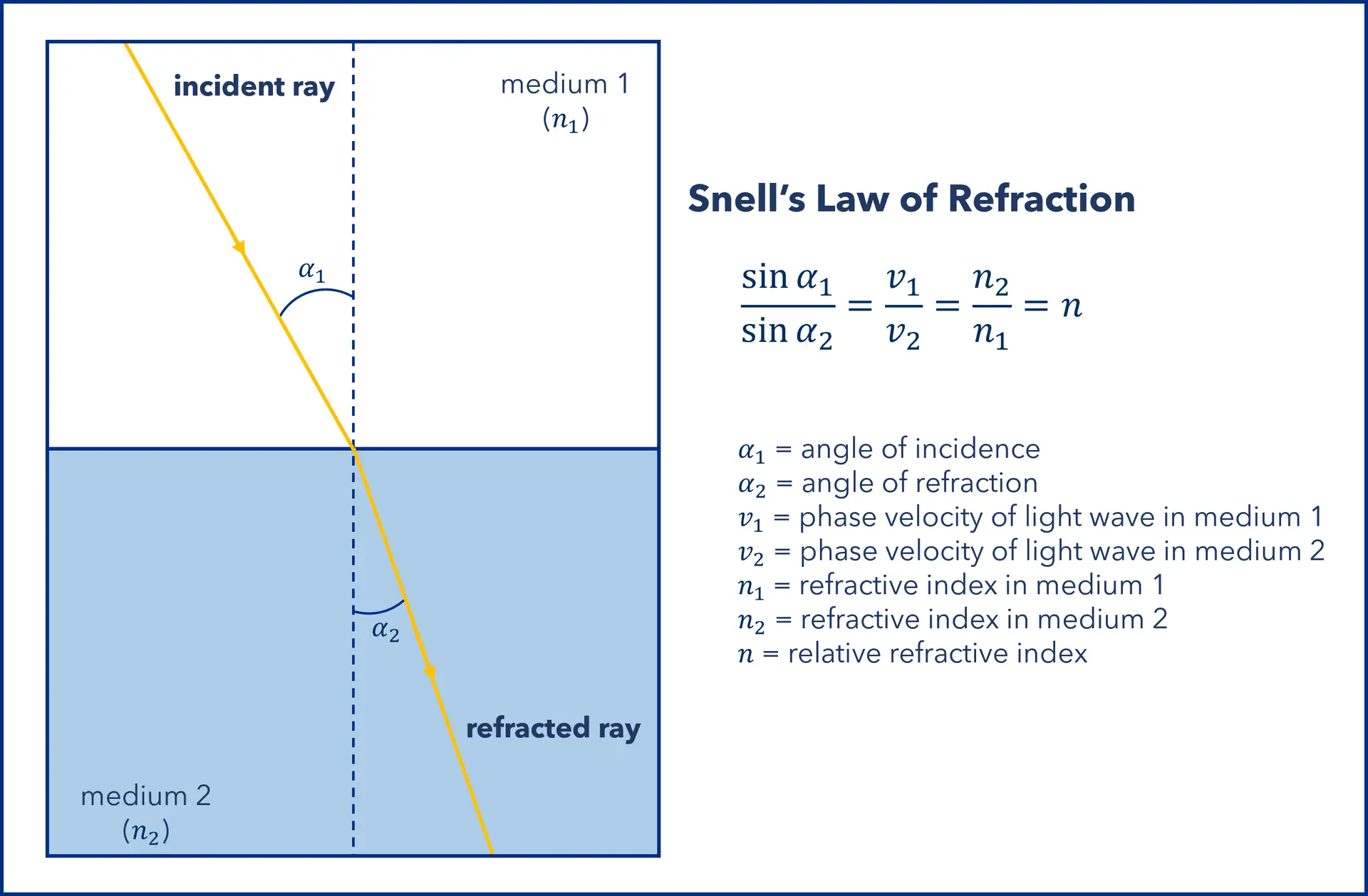
Figure 1: Refraction of light at the interface between two media with different refractive indices, where n₁ > n₂. Since the velocity is lower in the second medium (v₂ < v₁), the angle of refraction α₂ is less than the angle of incidence α₁.(Graphic by KNAUER)
In HPLC, an RI detector measures small differences in the refractive index of the mobile phase as an analyte elutes from the column. Since every compound has its own refractive index, even slight compositional changes in the eluent can be detected.
Modern RI detectors use a differential design (see Figure 2), continuously comparing two optical paths: one passing a reference cell containing pure mobile phase and one a sample cell containing column effluent (mobile phase + analyte). A light beam, typically from an LED or tungsten lamp, passes through both cells. A photodiode measures any deviation in the path as differences in refractive index occur.
When only mobile phase flows, both paths are optically identical. The light beam passes through without deviation, and no signal is recorded. When an analyte elutes, it changes the refractive index in the sample cell, slightly deflecting the beam and creating an optical imbalance. This shift results in a change in light intensity, which is detected and converted into an electrical signal that appears as a peak in the chromatogram. The signal response is proportional to analyte concentration, enabling quantitative analysis.
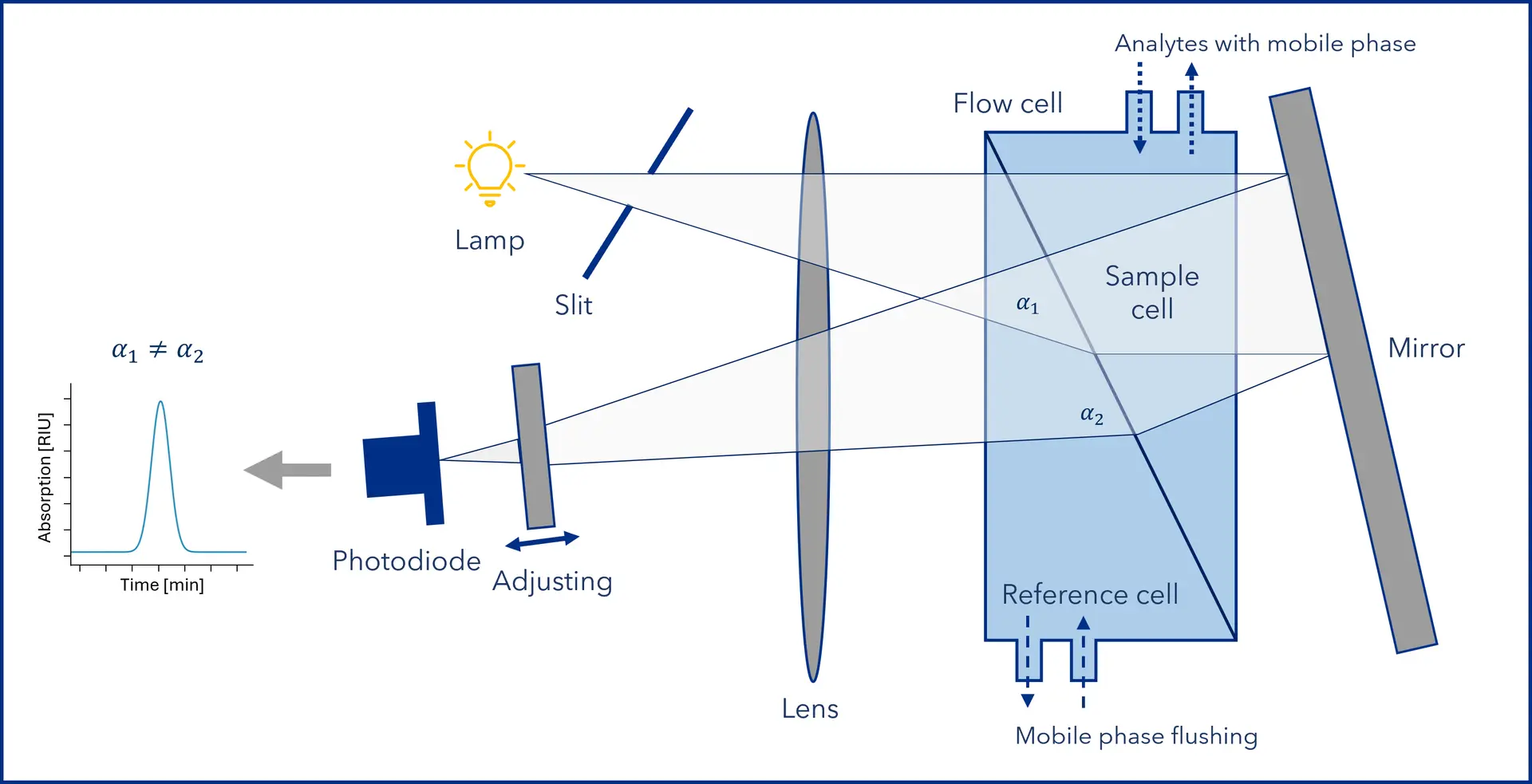
Figure 2: Schematic of a differential RI detector with dual flow cell. The sample cell carries column effluent; the reference cell contains pure mobile phase. A light beam passes through both and forms a slit image on a photodiode. Any analyte-induced change in refractive index alters the beam’s path, shifting the image on the photodiode and generating a signal peak. (Graphic by KNAUER)
To keep measurements stable, the reference cell must always be filled with the same mobile phase as the sample cell (without the analyte), so it is regularly refreshed with clean mobile phase, usually via a switching valve and/or T-piece (see Figure 3).
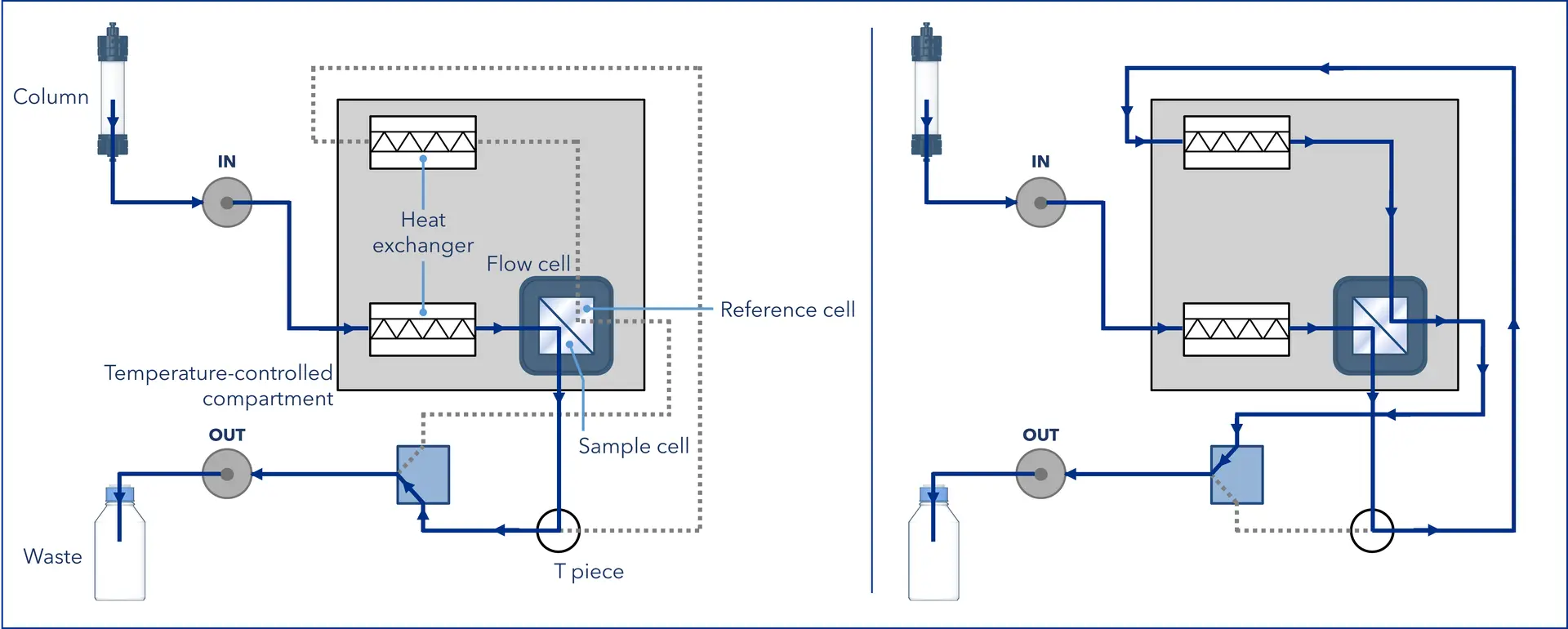
Figure 3: Flow path in the AZURA® RID 2.1L during analysis (left) and flushing (right). During analysis, solvent flows from the IN port through the heat exchanger and sample cell, then continues through the T piece and flush valve to the OUT port. During flushing, the valve redirects the flow through the T piece so the solvent passes a second heat exchanger and reference cell before exiting via the OUT port. (Graphic by KNAUER)
It’s worth noting that refractive index depends on both light wavelength and medium density. While the wavelength is fixed by the detector design, the density, and therefore the refractive index, varies by temperature, pressure, and composition. This makes RI detection highly sensitive to fluctuations in operating conditions. We’ll explore this topic in more detail in the following sections.
💡The Charm: Why Choose RI Detection?
RI detectors really shine when other detection modes fall short. They may not be the flashiest option, but they offer key advantages that make them a must for certain analytes.
🌍 True Universality
RIDs detect any analyte that changes the refractive index of the mobile phase – no chromophores needed. And because pretty much everything affects the refractive index, RI detection is called "universal detector". This means reliable detection of sugars, alcohols, lipids, fatty acids or polymers.
♻️ Non-Destructive Detection
RI detection leaves your sample intact, making RI a good fit for workflows where analyte recovery matters, such as preparative HPLC. Check out KNAUER’s AZURA® RID 2.1L for preparative applications up to 100 ml.
⚙️ Straightforward & Reliable
RI detectors work well with aqueous and many organic mobile phases, provided the composition is constant (hello, isocratic runs 👋). With stable conditions, they are pretty low-maintenance, robust, and easy to operate.
🧪 Direct Response
With RI detection, no derivatization or labeling is needed to make analytes "visible". As long as they are soluble in the mobile phase and affect the refractive index, they can be detected, keeping sample prep simple and reducing potential errors.
That simplicity makes RI detection great for routine analyses like quantifying lactose in milk or checking polymer purity.
💡The Catch: Limitations You Should Know
Every detector has its own quirks, and RI detectors are no exception. While they’re wonderfully universal, they demand stable and carefully controlled conditions to deliver their best performance, so they are a bit like the "divas" of HPLC labs.
Here are the main challenges.
🌡️ Temperature Sensitivity
RI detectors are extremely sensitive to temperature. Refractive index n varies linearly with temperature (dn/dT ≈ 10⁻⁴/°C for aqueous solvents), so even tiny fluctuations can cause noticeable baseline drift exceeding analyte signals.
That’s why RI detectors come with thermostated compartments and heat exchangers to tightly stabilize flow cell temperature. This allows for accurate and reproducible detection, but the heat exchangers also add some extra dead volume, which can broaden peaks compared with UV detection.
⚗️ Gradient Incompatibility
RI detection is essentially limited to isocratic runs. Any change in mobile phase composition shifts the refractive index far more than the analyte does, leading to baseline shifts that can hide peaks. A stable baseline depends on sample and reference cells containing the same mobile phase throughout the run.
🔍 Sensitivity
Compared to UV, RI is less sensitive, typically by 1-2 orders of magnitude. Detection limits are usually in the µg-mg/mL range, so it’s less ideal for trace analysis or impurity profiling.
💪 Pressure Sensitivity
RI flow cells tolerate only moderate backpressure, so when used in series, the RI detector must be placed last.
⏱️ Equilibration Time
RI detectors need time to stabilize. Warm-up and equilibration can take hours before a steady baseline is achieved.
📉 Baseline Instability
RI detection responds to any refractive index change. Even when the temperature is steady, flow and pressure fluctuations, air bubbles, pump pulsing, poor solvent mixing, or mobile phase impurities can all introduce noise or drift.
This makes clean and stable operation essential.
💡Getting the Best from Your RI Detector
Using an RI detector effectively comes down to one word: stability. Even tiny changes in operating conditions can quickly show up in your baseline and affect data quality, so a bit of care in RID setup and handling goes a long way.
🌡️ Keep Temperature Steady
Thermal stability is usually the first priority. Let the detector, column, and mobile phase fully equilibrate before starting a run for at least 60 min. Use a column oven and keep the detector and column at the same temperature whenever possible. Minimize environmental influences – drafts, sunlight, or day/night lab temperature swings can still throw things off.
📈 Stay Isocratic
RI detectors require constant mobile phase composition, so stick to isocratic methods. If your method relies on gradients, this detector simply isn’t the right tool.
🫧 Degas & Keep it Clean
Use fully degassed, particle-free mobile phases and make sure the system is properly primed. Make use of the in-line degasser in your LC system. Keep tubing, solvents, and flow path clean. Tiny air bubbles and impurities can cause spikes, noise, or false signals.
🧪 Prepare Samples Carefully
Dissolve samples in the mobile phase or in a solvent that closely matches it to avoid void time RI jumps. Always filter samples to reduce the risk of clogging and protect the flow cell.
💪 Stabilize Flow & Pressure
Keep flow rates constant and avoid sudden pressure changes, excessive pump pulsation, and leaks. A small restriction capillary or a back-pressure regulator installed after the cell can help stabilize pressure, but they won’t fix noise caused by pump issues.
⚙️ Maintenance, Interpretation & Practical Constraints
Flush the reference cell daily with fresh mobile phase, replace the mobile phase regularly to prevent microbial growth or composition changes due to evaporation, and avoid online mixing when possible.
When switching solvent systems, flush the instrument with mutually miscible solvents and allow enough time for re-equilibration. Using a mobile phase recycle mode or putting the detector in low flow standby between runs can help save solvent and reduce restart time.
👉 Pro Tip: Watch the baseline closely, a stable baseline is your best indicator that the system has equilibrated properly. And don’t be surprised by negative peaks; they simply mean the analyte has a lower refractive index than the mobile phase and are not necessarily a system error.
🎯 The Bottom Line
With careful attention to these details, an RI detector can deliver smooth baselines and reliable, consistent quantitation. Keep your system clean, your temperature stable, and your mobile phase consistent, and your "diva" will perform like a star.
💡Typical Applications: When RI Detection Shines
RI detection really comes into its own when analytes don’t absorb UV light, making it a go-to for compounds without strong chromophores, such as sugars, alcohols, fatty acids, polymers, lipids, and other small organic molecules.
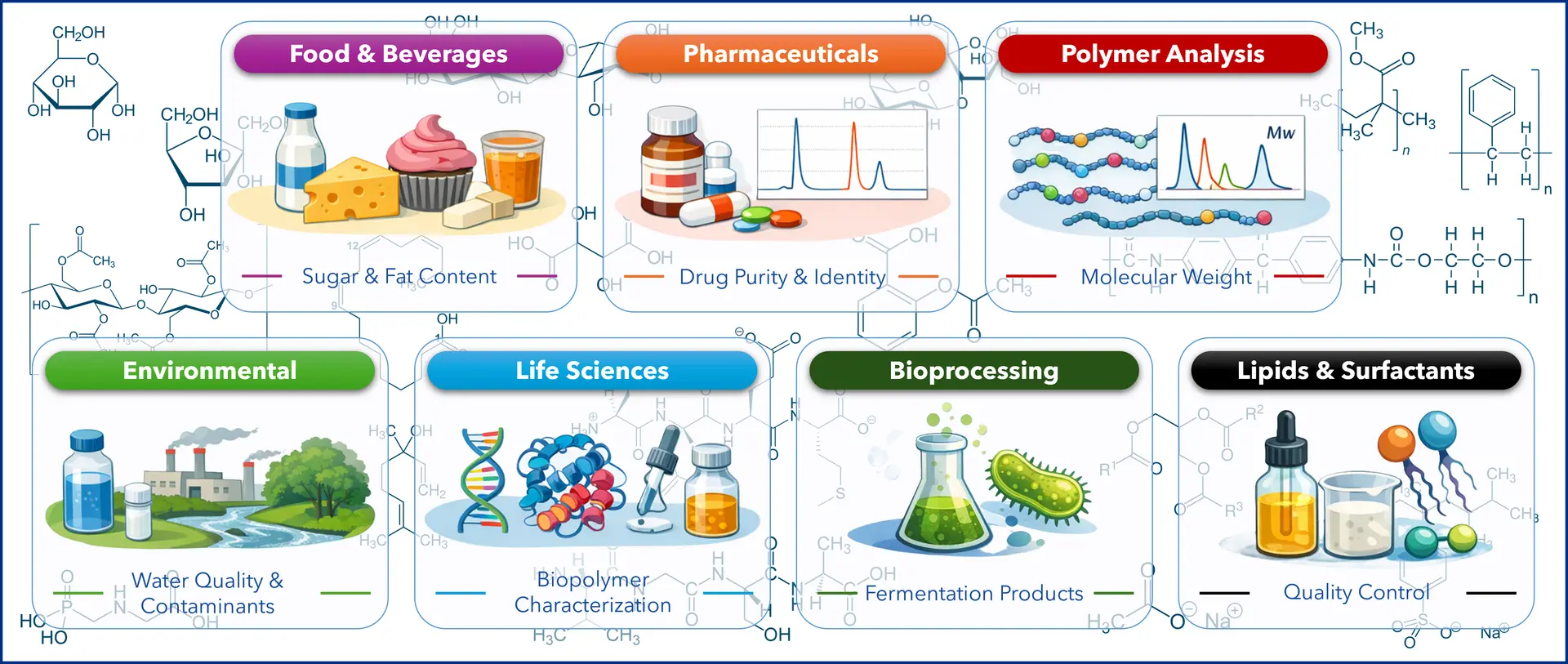
Figure 4: Common uses of RI detection in HPLC. (Graphic by KNAUER. Icons generated by DALL-E 3, OpenAI, GPT-5.4; April 10th, 2026)
Some typical applications include:
Carbohydrate and sugar analysis, e.g. tracking glucose, fructose, sucrose or lactose in food and beverages. Our colleague Julia recently used HPLC-RI for product quality control of gummy candies. You should also check out her blog post on the subject. But be warned, you might find yourself craving sweets while reading it 😉. She also used KNAUER’s AZURA® RID 2.1L refractive index detector to simultaneously determine sugars and organic acids in wine and revealed "The hidden science in your glass".
Polymer and macromolecule characterization in GPC/SEC workflows, such as determination of molecular weight distribution. Check out how molecular weight determination can be optimized using a KNAUER AZURA® GPC/SEC system or how mucin proteins can be analyzed using an AZURA® SEC System.
Bioprocessing, e.g. monitoring fermentation products like fructans.
Pharmaceutical applications, e.g. analysis of excipients such as sugars or polyols, or checking identity and purity of APIs. Dextranes, which are used as carrier substances in pharmaceuticals and as thickening and moisturising agents in cosmetics, are characterized by SEC, since their properties depend on a specific molecular weight distribution.
Lipid and surfactant analysis for quality control and formulation work.
Environmental analysis, e.g., detecting organic and inorganic impurities in water samples.
In short, RI detection works best when analytes are soluble, stable in solution, and run under simple isocratic conditions. While it is not suited for trace-level analysis, gradient methods, or MS-coupled workflows, it remains a reliable, practical solution for compounds that are difficult to detect with UV or fluorescence.
💡Wrapping Up: A Detector Worth Noticing
In a world of ever-more-advanced detection technologies, RI detection may not be the flashiest tool in HPLC, but it is one of the most dependable. For compounds like sugars, alcohols, and polymers, it offers a simple, reliable, and non-destructive way to detect what other methods may miss.
Its main limitations are temperature sensitivity and incompatibility with gradients, so it works best in carefully controlled isocratic methods. In the right setup, though, RI detection remains a practical lab workhorse – proving that sometimes, simplicity and universality are exactly what you need.
Pro tip: When your analyte seems "invisible", it might just be time to look at it differently. RI detection could be exactly what you need to bring it into view.
Have a look at KNAUER’s refractive index detectors here or contact our Sales team for help choosing the right detector for your needs.
In the next post of our "Seeing is Believing - Detection Made Easy" series, we’ll explore Fluorescence Detection (FLD), a highly selective and ultra-sensitive technique for compounds that fluoresce or can be made to.
For further information on this topic, please contact our author: huhmann@knauer.net
Resources
L. R. Snyder, J. J. Kirkland, J. W. Dolan, Introduction to Modern Liquid Chromatography, 3rd Edition, John Wiley & Sons, Inc., Hoboken, New Jersey, 2010.
D. A. Skoog, F. J. Holler, S. R. Crouch, Principles of Instrumental Analysis, 7th Edition, Cengage Learning, Boston, 2018.
R. P. W. Scott, Liquid Chromatography Detectors. In J. Chromatogr. Library, Vol. 11, Elsevier Scientific Publishing Company, Amsterdam, 1977, pp. iii-ix, 1-248.
V. R. Meyer, Praxis der Hochleistungs-Flüssigchromatographie, 10., vollst. überarb. u. erw. Auflage, Wiley-VCH, Weinheim, 2009.
G. Aced, H. J. Möckel, Liquidchromatographie, Apparative, theoretische und methodische Grundlagen der HPLC, VCH Verlagsgesellschaft mbH, Weinheim, 1991.
M. W. Dong, HPLC and UHPLC for Practicing Scientists, 2nd Edition, John Wiley & Sons, Inc., Hoboken, New Jersey, 2019.

