Science with Passion
Application No.: VBS0063
Version 1 03/2017
Automated two step purification of mouse antibody IgG1
Yannick Krauke, Kate Monks; applications@knauer.net
KNAUER Wissenschaftliche Geräte GmbH, Hegauer Weg 38, 14163 Berlin
Summary
This application highlights the possibility of automated purification of antibodies (IgG) with the AZURA® Bio purification system without manual interaction during purification process. The cell culture was applied with a feeding pump on a protein A affinity column to capture and purify the antibodies. These were kept in the system and in a second step applied on a gel filtration column for buffer exchange. Consequently, the obtained antibody was dissolved in the desired storage and/or working buffer.
Introduction
Antibodies (immunoglobulins, Ig’s) are part of the immune system. They can identify and bind particular antigens thereby neutralizing them. Due to their specific target recognition/binding function they have a significant importance in the biotechnology and pharmaceutical industry. Key applications are the diagnosis and treatment of diseases. Besides, antibodies are also the crucial components in numerous research applications such as Western Blots and immunoassays. Quality and purity of the IgG is crucial for these applications. The purification of antibodies involves two to three steps, 1. capture step, (2. intermediated step), 3. polishing step. The transition from one to another step generally involves manual interaction and thus is time consuming. The aim of this application note was to establish an automated purification method on the AZURA Bio purification system combining an affinity chromatography step with a gel filtration/desalting step to exchange the buffer of the purified antibodies.
Results
The mouse immunoglobulin (IgG1) was purified from 10 mL cell culture by affinity chromatography, using a protein A column. The chromatogram of the IgG purification shows the four main phases of the procedure (Fig.1). Phase 1: equilibration of the protein A column with buffer A . Phase 2: sample injection by the feed pump. The large flow through peak (A) visualizes the cell culture matrix and proteins not bound by the protein A column. Subsequently, the column was washed with buffer A until no further peaks were detected. Phase 3: elution of the captured IgG1 with buffer B and parking in the sample loop (B1). Phase 4: immediate buffer exchange was performed by the flushing of the system with exchange buffer C and the following re-injection of the IgG1 on the desalting column. The eluting peak was recovered by the fraction collector (B2). The main aim of the second step was the buffer exchange. The conductivity signal was recorded, demonstrating the desalting of the eluates during the purification process (Fig. 2). Finally, a SDS-PAGE was performed to control the result of the purification steps (Fig. 2). The analysis of the flow through and comparison with the injected sample show that some IgG1 did not bind to the protein A column (lane 1 and 2). The protein bands of IgG1 heavy chains (HC) and IgG1 light chains (LC) are visible at 55 kDa and 22 kDa in the SDS-PAGE. Further, a larger un-specific band at 70 kDa was detected. This band was only detected in the flow through and not in the purified IgG (lane 2, 3, 4) showing that IgG1 was not contaminated with other proteins. The IgG1 after the protein A column (lane 3) and after the desalting column (lane 4) have a similar concentration showing no protein loss in the second purification step.
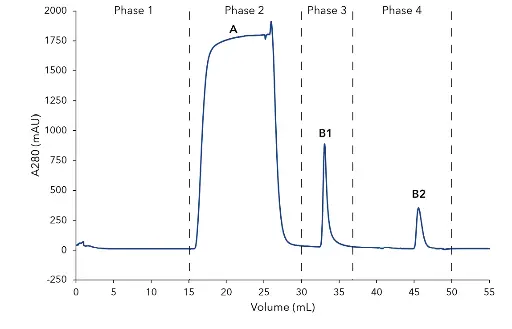
Fig. 1 Chromatogram of the two-step mouse IgG purification; Phase 1 Affinity chromatography (AC): 1 Column equilibration with buffer A; 2 Feed injection and column washing; 3 Elution of IgG from protein A column with buffer B and parking in 1mL sample loop; Phase 2 - Buffer exchange with desalting column: 4. Elution of IgG with buffer C; A–flow through; B1–elution peak of IgG from protein A column; B2-elution peak of IgG after desalting column
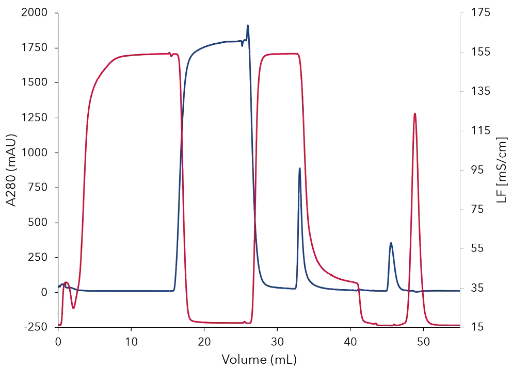
Fig. 2 Chromatogram of the two step mouse IgG purification with UV and conductivity signal; UV signal at 280 nm in blue; conductivity sign al in red; after desalting (45min) elution peak (blue) and salt peak (red) are clearly separated
Materials and Method
The AZURA two step purification system was used for this application. It consists of AZURA P 6.1L HPG; 1st ASM 2.1L with feed pump and two 6 port/3 channel injection valves; second ASM 2.1L with UVD 2.1S and two 6 port/3 channel injection valve; a column switching valve; a conductivity monitor and a fraction collector. The protein A column (ZetaCell protein A , 1ml) was equilibrated with 15 ml buffer A (TBS) at 1 mL/min. Then 10 mL of feed at 1mL/min were injected and column washed with 5 mL buffer A at 1 mL/min. The antibody was then eluted with 10 mL elution buffer B (0.2 M Na Citrate, pH 3) and the eluting antibody collected in sample loop. Finally, the protein was re-injected and eluted with 15 mL of buffer C (PBS pH 7.4) over the desalting/gel filtration column at 1mL/min. The UV signal (280 nm) and the conductivity signal were recorded.
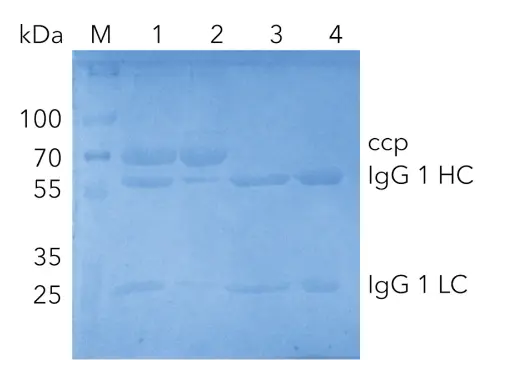
Fig. 3 SDS-PAGE of IgG1 at different purification steps;
M) Marker in kDa; 1) cell culture supernatant; 2) flow through; 3) pure IgG1 after elution from protein A column;
4) pure IgG1 after desalting column; ccp cell culture proteins; IgG1 HC (heavy chain); IgG1 LC (light chain)
Conclusion
Mouse IgG was successfully purified from cell culture medium by an automated combination of an affinity chromatography and gel filtration method on the two-step dedicated AZURA Bio purification system. No manual interaction was necessary. The method setup could easily be adapted to other purification protocols for the separation of biomolecules. This application is an example of a time-saving automation of protein purification and can be easily adapted to various protein purification protocols.

Additional Materials and Methods
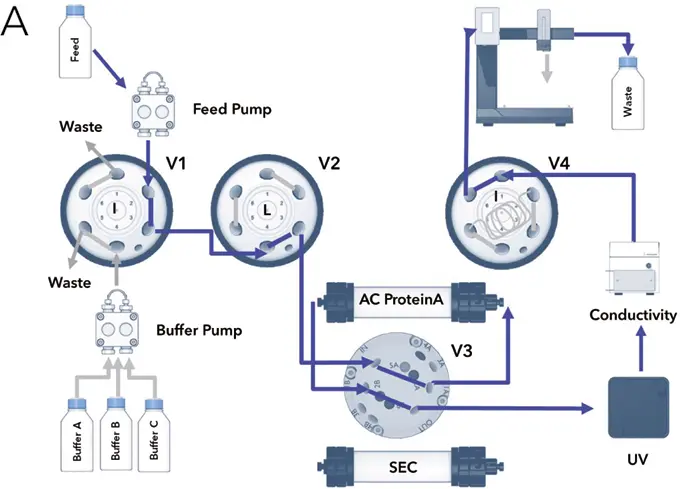
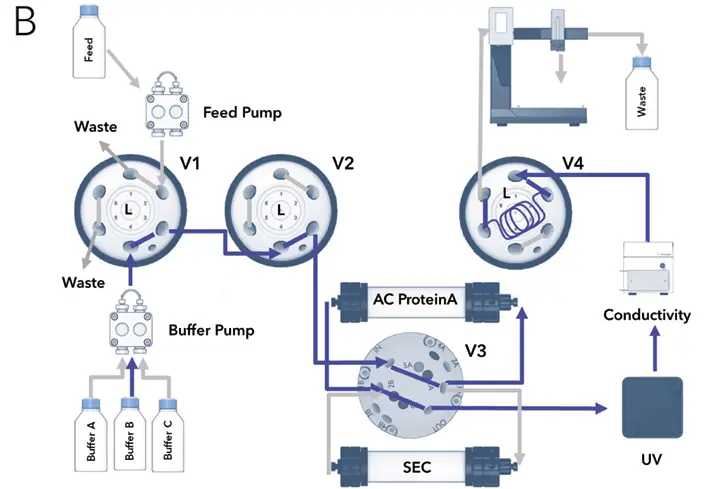
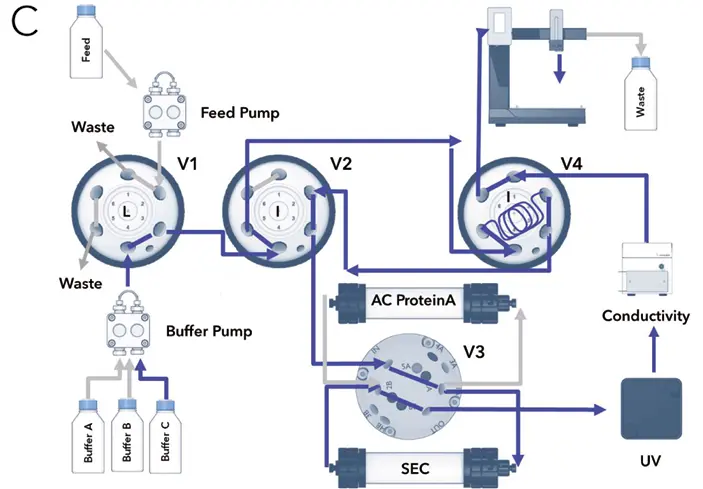
Fig. A1 Flow chart of the two – step purification illustrating major method steps; A Feed injection and antibody binding on protein A column, flow through to waste, washing afterwards with buffer A (not shown); B Protein elution from protein A column with buffer B and protein parking in sample loop; C Re-injection of parked protein on SEC desalting column, buffer and pH exchange using buffer C, fractionation of target peak; valve positions = I) injection, L) load
Fig. A2 The system configuration and the different valve settings are shown in Fig. A1. The first injection valve (V1) is used for the injection of the sample. To this valve the feed pump and the buffer pump are connected. The other two injection valves are necessary for the inversion of the flow direction (V2 + V4). They also switch the flow to the waste/fraction collector and in/out of the sample loop. The sample loop allows the collection and (re)injection of the peak of interest. The two columns are connected the to the column switching valve (V3). In the initial configuration (Fig. A1 A) the sample is injected onto the column by the feed pump. The non-binding protein is directed to the waste. After washing the column, IgG is eluted with eluent B (Fig. A1 B). The valves are switched to the peak parking position (V2 + V4 in Load position). The eluting protein is collected in the sample loop. Subsequently, the collected protein is automatically re-injected onto the second column by changing the valve position for V2 and V4 from Load to Inject and for V3 from position 1 (column 1) to position 2 (column 2) (Fig. A1 C). The flow is inverted and the sample loop is emptied. The eluted protein peak is fractionated by a fraction collector. No manual interaction is necessary during the purification.
Tab. A1 Method parameters
Tab. A2 2 Purification steps run at a flowrate of 1 mL/min; AC - affinity chromatography, GF - gel filtration

AZURA® Two Step Purification System
Tab. A3 System configuration & data
Anwendungsdetails