
Science with Passion
Application No.: VPH0066
Version 1 04/2019
Cyclodextrin purification (Part 1): Method screening and overload studies
Yannick Krauke*, Sebastian Thürmann*, Christian Sonnendecker**, Kate Monks*; applications@knauer.net
*KNAUER Wissenschaftliche Geräte GmbH, Hegauer Weg 38, 14163 Berlin
** Fakultät für Lebenswissenschaften/Institut für Biochemie AG Mikrobiologie und Bioverfahrenstechnik,
Johannisallee 21-23, 04103 Leipzig
Summary
Cyclodextrins (CD) are macrocyclic compounds composed of five or more glycopyranosides. These ring structures can function as micro-capsules. An automated column screening was applied for method development to find best separation conditions of a CD mixture. Finally, an optimized method for CD purification was established.
Introduction
Cyclodextrins (CD) are oligosaccharides of glucopyranose that are bound in a cyclic form of six to at least 12 units. The ring structure can function as mirco-capsule for other molecules. Do to their unique chemical structure they can find different applications i. e. as drug carriers, in cosmetics or in food industry. CDs with more then 10 subunits are of special interest as larger molecules can be inserted in these rings. Therefore, the approaches for synthesis and purification of CD >10 are conducted [1]. Here, a method optimization was performed for the purification of cyclodextrins from biocatalytic synthesis [2]. With an automated column screening an existing method was optimized. Further, mass- and volume overload studies were performed in analytical scale to find best conditions for scale-up to preparative scale.
Results
A device with automated column switching valves was used for sequentially testing of four different stationary phases: C18, C18H, C18A und C18P each 150 x4 mm, 5 µm particles and 2.5 % methanol (Fig. A1, additional results). The C18 and C18H showed the best separation profile among tested columns. Separation with water on aqueous C18A and C18AP revealed no promising results.
Next, the columns length was investigated and revealed that the relevant peaks had a better separation factor on a 250 mm then 150 mm long columns (Fig. A2, additional results). The 150 mm length column is good for analysis but for preparative applications the 250 mm column is required.
Comparison of different methanol concentrations revealed that 5 % methanol was too high as nearly all peaks eluted together within the first 10 min (not shown). At 3 % methanol one additional peak (Fig. A3; CD13, additional results) was detected which was not found at 2.5 % methanol on C18 column (Fig. A3), revealing that accurate eluent preparation is essential for this method.
The final separation profile comparison of the CDmix on C18 and C18H revealed that the C18H column is the better choice for purification of cyclodextrins due to earlier elution of target peaks (Fig. 1). Four relevant cyclodextrins were baseline separated (Fig. 1). A mass and volume overload studies were performed prior to preparative scale-up. Four different concentrations (25, 50, 75, 100 mg/mL) of the CD mix were tested and results showed that even at 100 mg/mL CD10, CD11 and CD12 were baseline separated (Fig. 2). Next, different volumes (50, 75, 100, 200 µL) of 50 mg/mL CD mix were injected. At 100 µL the first three peaks were still baseline separated, at 200 µL not as well anymore (Fig. 3). The resulting data was used to up-scale the separation for purification on columns with larger ID (see application Cyclodextrine purification – VPH0068).
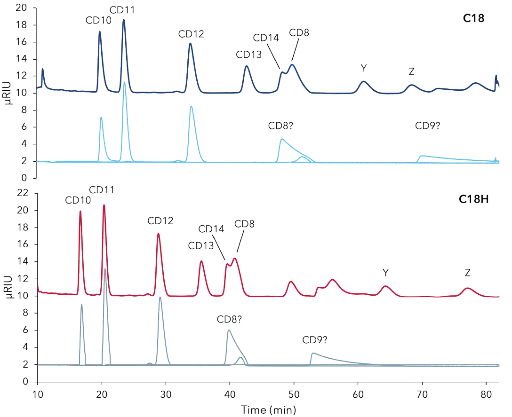
Fig. 1 Comparison of separation profiles from CD mixture and CD standards on C18 (dark blue) and C18H (red) columns (relevant time span of separation shown). CD mixture (30 mg/mL), CD8 (1mg/mL), CD 12,9,8 mixture (3mg/mL), CD10, 11 mixture (2 mg/mL); Y, Z unidentified peaks; 250x4mm, 5 µm, 0.8 mL/min 3 % methanol, 50 µL inject
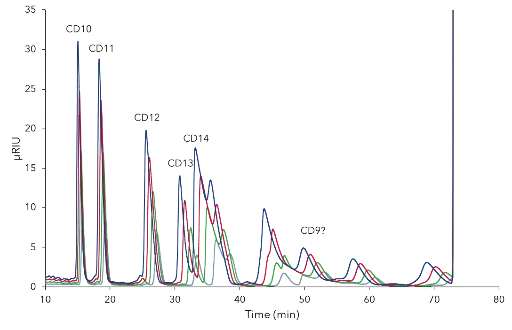
Fig. 2 Chromatograms of CD mixture mass overload studies; grey – 25 mg/mL, green – 50 mg/mL, red – 75 mg/mL, dark blue – 100 mg/mL; C18 H 250 x 4 mm; 5 µm; 0.8 mL/min; 3 % methanol; 25°C; 50 µL
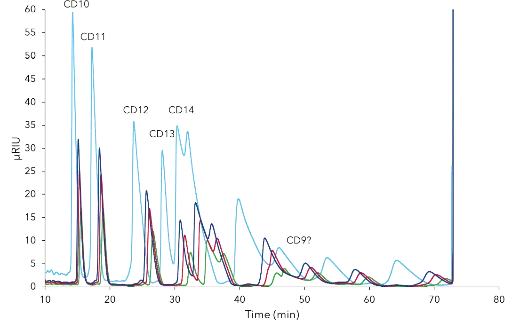
Fig. 3 Chromatograms of CDmix volume overload studies. green – 50 µL, red – 75 µL, dark blue – 100 µL, light blue – 200 µL; C18H 250 x 4 mm; 5 µm; 0.8 mL/min; 3 % ,methanol; 25°C; 50 mg/mL
Materials and Method
The AZURA HPLC system consisted of AZURA P 6.1L 10 ml HPG sst pump, AZURA AS 6.1L autosampler, AZURA RID 2.1L detector, AZURA Assistant with two 6 port multi-position stainless steel valves for column switching and an AZURA CT 2.1 column thermostat. Final method was as follows: 0.8 mL/min, 25°C, 70 min at 3 % methanol, 10 min at 30 % methanol, 40 min at 3 % methanol.
Conclusion
An automated column switching assistant is the optimal device for fast and effective stationary and mobile phase screening. An existing method was optimized with focus on a later up-scaling for purification of CDs. The method will allow to purify CD10, CD11 and CD12 in high purity in batch purification process.
Additional Results
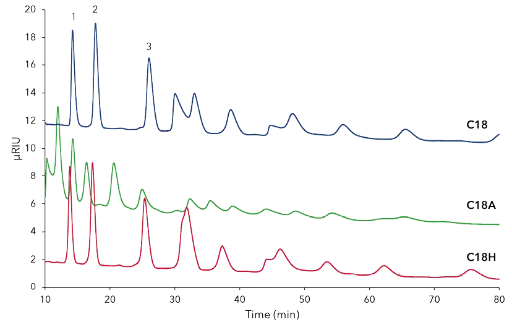
Fig. A1 Comparison separation of CD mixture (25 mg/mL) on three different stationary phases; blue – C18, green – C18A, red – C18H; 1 – CD10, 2 – CD11, 3 - CD12; all columns 150 x 4mm, 5 µm, 0.8 mL/min, 25°C, 50 µL inject.
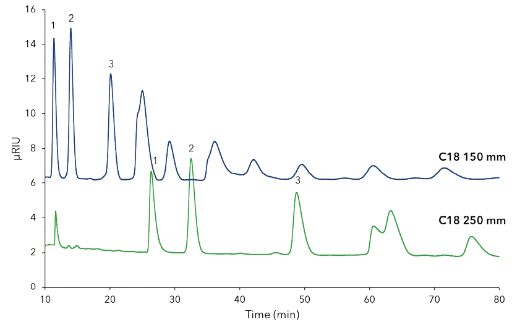
Fig. A2 Comparison separation of CDmix (25 mg/mL) on C18 columns with different lengths; CDmix (25 mg/mL), blue -150 x 4 mm, green - 250 x 4 mm; 1 – CD10, 2 – CD11, 3-CD12, 5 µm, 0.8 mL/min, 25°C, 50 µL inject
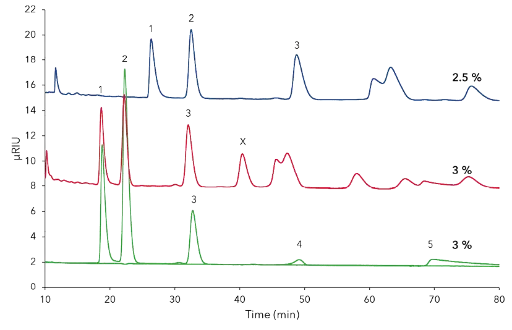
Fig. A3 Comparison separation of CDmix with 2.5 % and 3 % methanol on C18 column 250 x 4 mm. CDmix (25 mg/mL), blue – 2.5 % methanol, red – 3.0 %, green - standards at 3.0 % methanol; 1) CD10, 2) CD11, 3) CD12, 4) CD8, 5) CD 9; 5 µm; 0.8 mL/min; 25°C; 50 µL
Additional Materials and Methods
Tab. A1 Method parameters
Tab. A2 Pump parameters
Tab. A3 System configuration

References
[1] E.M. Martin del Valle (2003), Cyclodextrins and their uses: a review, Process Biochemistry, Volume 39, Issue 9, Pages 1033-1046, ISSN 1359-5113
[2] Sonnendecker, C. , Thürmann, S. , Przybylski, C. , Zitzmann, F. D., Heinke, N. , Krauke, Y. , Monks, K. , Robitzki, A. A., Belder, D. and Zimmermann, W. (2019), Large‐Ring Cyclodextrins as Chiral Selectors for Enantiomeric Pharmaceuticals. Angew. Chem. Int. Ed.. doi:10.1002/anie.201900911
Related KNAUER Applications
VPH0068 – Cyclodextrin purification (Part 2): Method transfer and purification
Application details


