
Science with Passion
Application No.: VPH0073
Version 1 06/2020
Purify CBD and other cannabinoids by preparative HPLC
Yannick Krauke, Kate Monks; applications@knauer.net
KNAUER Wissenschaftliche Geräte GmbH, Hegauer Weg 38, 14163 Berlin

Summary
The demand of pure cannabidiol (CBD) and other cannabinoids is increasing and therefore methods are needed that allow simultaneous purification of several cannabinoids from the same sample i.e. cannabis extract, paste or oil. Preparative HPLC is a well-established method for purification of target substances from natural substrates in high purities. In this application, a preparative HPLC method was developed for purification of CBD, Δ9-THC and CBC from a commercial CBD rich oil. All three cannabinoids were purified with high purity thus increasing the purity of the main component CBD from 72% to 93%.
Introduction
Cannabis sativa is used in agricultural and medical applications for already long time. It contains a variety of compounds such as terpenoids, flavonoids and cannabinoids among which tetrahydrocannabinol (Δ9-THC) and cannabidiol (CBD) are the most abundant1. Δ9-THC is known for its psychoactive effects and is applied as medical treatment, but underlies strict legal regulations in most countries. CBD does not have the intoxicating effects of THC, and therefore the legislations are much more relaxed, yet. Depending on the field of application there are cannabis breeds with high THC content and others with low THC but high CBD content. As CBD shows medicinal effects as THC and is used i.e. for the treatment of neuropathic pain and chronic inflammatory conditions, the demand is growing. Many products such as CBD oil, drops and cosmetics are also available on the market2,3,4,5. The demand of highly pure CBD and other cannabinoids is increasing, and therefore different purification strategies are developed. If the goal is to purify CBD from a sample, in most cases a pre-processed paste from CBD rich hemp, besides preparative HPLC flash or centrifugal partition chromatography (CPC) could be applied. If the aim is to purify several different cannabinoids from the same sample, preparative HPLC has its advantages. Here the purification of CBD, Δ9-THC and CBC (cannabichromen) from a CBD oil using a KNAUER AZURA preparative HPLC system is described. Further, the whole process from sample analysis to method development in analytical scale, the scale up to preparative scale and finally the purification of target cannabinoids is shown. Due to strict regulatory conditions concerning the novel food legislation in 2019, it was difficult to acquire a CBD rich paste or other CBD process sample. Exemplarily, a commercial CBD rich oil was used as sample.
Results
The CBD–Loges oil was analyzed with the previously described acetonitrile method6. The CBD oil chromatogram showed one large peak at tR 8.9 min and several significant smaller peaks (Fig. 1).
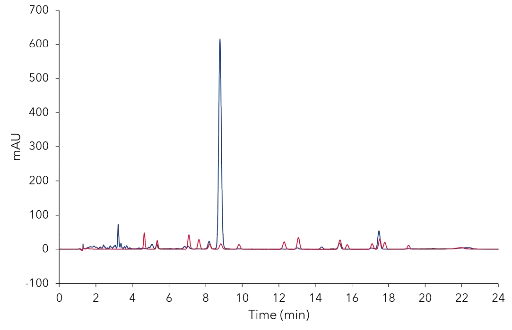
Fig. 1 Overlay chromatograms of CBD oil sample (blue) and mixture of 16 cannabinoids (red). CBD oil sample 5 mg/ml; standard concentration
5 µg/ml each; 10 µl injection, 228 nm; C18P, 3 µm, 150 x 4.6 mm ID.
Most impurities eluted within the first five minutes (Fig. 2). An overlay of the chromatograms of a standard mixture and the mixture of 16 cannabinoids, as well as a subsequent retention time comparison led to the identification of seven cannabinoids: CBDV, CBG, CBD, CBN, Δ9-THC, CBL and CBC. A larger, early eluting peak group eluted within the first 5 minutes, and two larger unidentified peaks at approximately 5 and 14.4 minutes (Fig. 2).
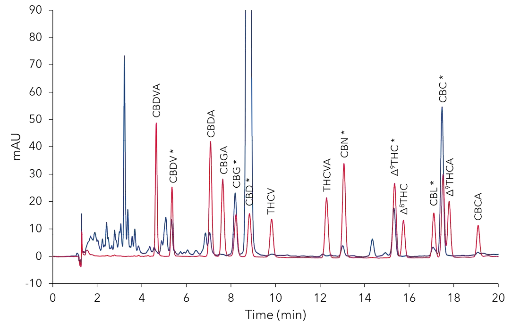
Detailed view of Fig. 1 with the 16 cannabinoids standard mixture. Positive identified substance peaks are highlighted with * in sample.
A five-point calibration curve for the seven cannabinoids was measured and the resulting coefficients were above R>0.999 for all seven cannabinoids (Fig. 3).
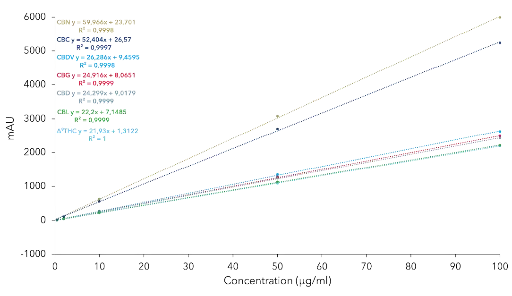
Calibration curves and linearity for the cannabinoids CBDV, CBG, CBD, CBN, Δ9-THC and CBL for analytical ACN method at 228 nm.
The calibration was used for the analysis of the CBD oil sample. The area % was used to quantify the purity of the identified cannabinoids. The results show that CBD is the most abundant cannabinoid in the sample with approximately 73% (area) followed by CBC with 5.7% (area). In the integration interval 15.6% (area) of the sample were unidentified peaks, other cannabinoids and impurities (Tab. 1). The calculated cannabinoids percentages in 1 mg CBD oil are 5.09% CBD and 0.17% Δ9THC which are close to the manufacture indications (5.35% CBD and <0.2% Δ9-THC). Content of CBC (0.18%) and CBG (0.17%) are similar to Δ9-THC (Tab. 1).
Analysis composition of identified cannabinoids in CBD oil sample (5 mg/ml). Integration interval 1.5 min – 20 min; global width 0.1 min; global threshold 0.1 mAU.
Area % | Concentration ug/ml | % in 1 mg CBD oil | |
CBDV | 1.00 | 2.86 | 0.06 |
CBG | 2.50 | 8.49 | 0.17 |
CBD | 71.90 | 254.34 | 5.09 |
CBN | 0.60 | 0.40 | 0.01 |
Δ9-THC | 2.20 | 8.36 | 0.17 |
CBL | 0.50 | 1.92 | 0.04 |
CBC | 5.70 | 8.88 | 0.18 |
others | 15.60 |
After the analysis of the sample, a purification method was developed to purify CBD with higher purity. Additionally, Δ9-THC and CBC should be purified with the same method to show the possibility to purify several different cannabinoids from one sample in one run. A fast methanol gradient method was developed in analytical scale with a total run time of 13 minutes (Fig. 4; Tab. 5).
CBD oil sample (5 mg/ml) with optimized methanol gradient method at 228 nm (blue) and 306 nm (red); C18, 3 µm, 150 x 4.6 mm ID.
Comparison of the chromatograms of the CBD oil sample and six cannabinoid standard chromatograms allowed the mapping of those (Fig. 5). CBG and CBD are nearly coeluting. Therefore purified CBD will contain always CBG using this method. The four other identified cannabinoids were eluting close to each other, but a distinct separation of Δ9-THC and CBC looked promising (Fig. 5).
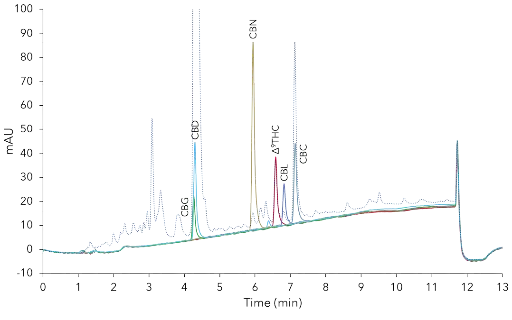
Detailed view overlay of CBD oil sample and single standard chromatograms. CBD-Loges oil (dashed line), standards in elution order: CBG
(5 µg/ml), CBD (10 µg/ml), CBN (10 µg/ml), Δ9-THC (10 µg/ml); CBL (5 µg/ml), CBC (5µg/ml), C18, 3 µm, 150 x 4.6 mm ID.
The optimized method was transferred to a C18 column with similar length and inner diameter but larger particle size (10 µm), which correspond to the particle size of the later preparative column. The maximum solubility of the oil in methanol/water (75%/25% v/v) was 50 mg/ml. Larger quantities were not soluble (data not shown). A volume overload study with a 50 mg/ml CBD oil sample was performed on this column. The results showed that even with 15 µl injections the CBD peak had nearly linear absorption behaviour (Fig. 6).
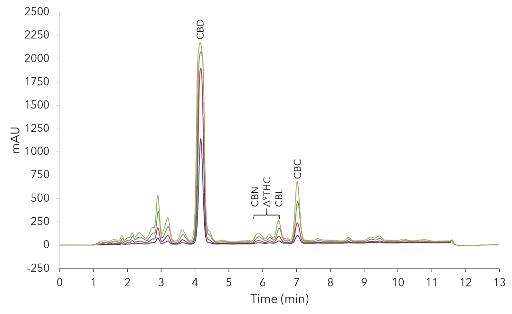
Volume overload study with methanol method on C18 10 µm particle. 2 µl, 5 µl, 10 µl, 15 µl injection of 50 mg/ml CBD-Loges sample; C18,
10 µm, 150 x 4.6 mm ID.
The increase of particle size slightly decreased the resolution, but it was still suitable for purification of the target compounds. A linear up-scaling of the method was performed. The column inner diameter (ID) was increased from 4.6 mm to 20 mm. The flow rate was increased from 1.2 ml/min to 23 ml/min. Parameters were calculated with the KNAUER ScaleUp Converter. Different injection volumes were tested starting from 200 µl up to 2000 µl of 50 mg/ml CBD oil sample. From the 2 ml injection run five fractions were collected with the aim to purify CBD/CBG, Δ9-THC and CBC (Fig. 7).
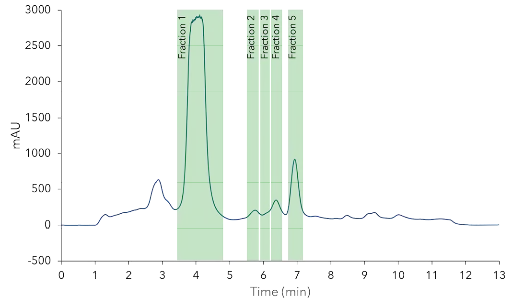
Preparative purification of CBD. 2 ml sample 50 mg/ml injection and fractionation; C18,10 µm, 150 x 20 mm ID.
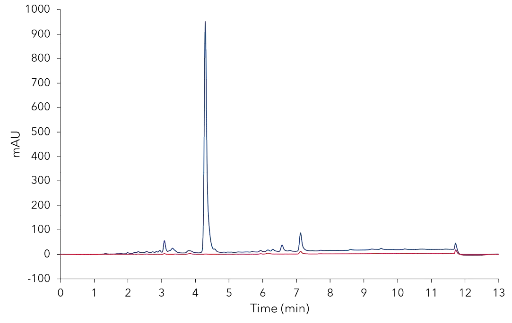
Automated fractionation was carried out by signal threshold of the PurityChrom® Software. The collected fractions were analyzed and the chromatograms were compared to the chromatogram of the injected CBD oil sample. The results clearly showed that only CBD, CBG and small amount of impurities were detected in fraction 1, but none of the early and late eluting compounds (Fig. 8).

Analysis of fraction 1 (red) from Fig. 7 and overlay with sample chromatogram (blue). 10 µl injection; analytical ACN method.
Whereas in fraction 5 mainly CBC is present (Fig. 9).

Analysis of fraction 5 (red) from Fig. 7 and overlay with sample chromatogram (blue). 10 µl injection; analytical ACN method.
The other fractions are not shown. The area percentages (indicator for purity) of the tested cannabinoids and the percentage of the total amount were determined for the five collected fractions (Tab. 2). In fraction 1 all of CBG and CBD were collected. The purity of CBD was increased from approximately 72% in the CBD oil to 93% in fraction 1. The fractions 2 and 3 contained a mixture of different cannabinoids but also high amounts >65% (area %) of unidentified compounds. The fraction 4 contained mainly Δ9-THC (~ 80%) and most of it from the sample (~ 90%). The fraction 5 contained 100% of the CBC with purity of approximately 97% (Tab. 2).
Analysis of fractions from preparative purification (2 ml injection with 50 mg/ml) of cannabinoids from CBD oil sample. For each fraction area % of identified cannabinoids and % from total sample are shown. Integration interval 1.5 min – 20 min; global width 0.1 min; global threshold 0.1 mAU.

Sample Preparations
A commercially available CBD rich oil was used for the application (CBD-Loges, 10 ml, with hemp extract containing 5.35% CBD). This oil contained also high amount of sesame oil. The oil was diluted in methanol to indicated concentration (i.e. 5 mg/ml) and sonicated for 15 minutes. After 10 minutes of settling the solution was filtered through 0.2 µm PTFE syringe filter to remove the unpolar part of the mixture. For higher oil concentrations (50 mg/ml) filtration and settling were repeated. Cannabinoid calibration standards were prepared from 1 mg/ml cannabinoid standard solution (Tab. 3) and diluted with methanol to indicated concentrations. The standards were prepared in two mix solution, mix 1 (CBDV, CBG, CBD, CBN) and mix 2 (Δ9-THC, CBL, CBC). A five-point calibration curve with 0.4 µg/ml, 2 µg/ml, 10 µg/ml, 50 µg/ml, 100 µg/ml of each of the seven identified cannabinoids was established.
Conclusion
The analysis of the CBD-Loges oil showed that the main cannabinoid was CBD with a 72% purity. The manufacturer’s information that the oil contains 5.35% CBD and <0.2% Δ9-THC could be confirmed with results from analysis (5.09% CBD and 0.17% Δ9-THC). A preparative methanol based step-gradient method was developed to purify CBD, Δ9-THC and CBC in high purities from the CBD oil. CBD was purified with 100% recovery resulting in a yield of 5.09 mg CBD from a 100 mg CBD oil injection ( 2 ml of 50 mg/ml solution). The results proved the simplicity of a linear scale up and the advantages to perform method optimization in analytical scale. This approach saves time, sample and solvent. The shown method is an example and was targeted at three cannabinoids. Depending on the target cannabinoids, the method could be adapted. It is important to mention that the yield could be significantly improved by using a sample with higher concentrations of the target compounds. Nevertheless, the same KNAUER AZURA preparative HPLC system could be used. Also, changing the solvent to ethanol, acetonitrile or even normal phase could be possible, but then the method development has to be started from the beginning.
KNAUER does not endorse the use of its products in connection with the illegal use, cultivation or trade of cannabis products. KNAUER does not endorse the illicit use of marijuana, we merely provide an overview of the methods and systems of cannabis analysis and purification.
Materials and Methods
Tab. 3 Standard solutions and samples
Analyte | CAS | Purity / Concentration |
Cannabichromene (CBC) | 20675-51-8 | 0.972 mg/ml (MeOH) |
Cannabidivarin (CBDV) | 24274-48-4 | 0.986 mg/ml (MeOH) |
Cannabidiol (CBD) | 13956-29-1 | 1.000 mg/ml (MeOH) |
Cannabigerol (CBG) | 25654-31-3 | 0.995 mg/ml (MeOH) |
Cannabinol (CBN) | 521-35-7 | 1.000 mg/ml (MeOH) |
Cannabicyclol (CBL) | 21366-63-2 | 0.992 mg/ml (ACN) |
Δ9-Tetrahydrocannabinol (Δ9-THC) | 1972-08-3 | 1.001 mg/ml (MeOH) |
Solvent | CAS | Purity / Concentration |
Acetonitril | 75-05-8 | Gradient grade |
H3PO4 | 7664-38-2 | AnalaR 85% NORMAPUR |
Methanol | 67-56-1 | Gradient grade |
Sample | Manufacturer | Batch |
CBD–Loges (Cannabis oil) | Dr. Loges | MA-19160911-2 |
Tab. 4 Analytical acetonitril method
Column temperature | 25 °C |
Injection volume | 10 µl |
Injection mode | Partial loop |
Detection | UV 225 nm/306 nm |
Data rate | 10 Hz |
Gradient | |||
Eluent (A) | Water, HPLC grade pH 2.2 (H3PO4 85%) | ||
Eluent (B) | Acetonitrile, gradient grade | ||
Flow rate | 1.0 ml/min | ||
Time (min) | A (%) | B (%) | |
Pump program | 0 | 25 | 75 |
7 | 25 | 75 | |
17 | 10 | 90 | |
19 | 10 | 90 | |
20 | 25 | 75 | |
25 | 25 | 75 |
Analytical and preparative gradient methanol method
Analytical | Preparative | ||
Column temperature | 25 °C | RT | |
Injection volume | x µl | 2000 µl | |
Injection mode | Partial Loop | Full loop | |
Detection | UV 228 nm / 306 nm | UV 228 nm | |
Data rate | 10 Hz | 2 Hz | |
Gradient | |||
Eluent (A) | Water | ||
Eluent (B) | Methanol, Gradient grade | ||
Flow rate | 1.2 ml/min (analytical) | 23 ml/min (preparative) | |
Time (min) | A (%) | B (%) | |
Pump program | 0 | 15 | 85 |
8 | 0 | 100 | |
10 | 0 | 100 | |
10.02 | 15 | 85 | |
13 | 15 | 85 |
Tab. 6 Analytical system configuration
Instrument | Description | Article No. |
Pump | AZURA P 6.1L LPG | |
Autosampler | AZURA AS 6.1L, 700 bar | |
Detector | AZURA DAD 2.1L | |
Puls Damper | High volume, stainless steel | |
Flow cell | Light guide 10mm/10 µl/300 bar | |
Thermostat | AZURA CT 2.1 | |
Column | Eurospher II 100-3 C18P, 150 x 4.6 mm ID | |
Column | Eurospher II 100-3 C18, 150 x 4.6 mm ID | |
Column | Eurospher II 100-10 C18, 150 x 4.6 mm ID | |
Software | ClarityChrom 8.1 – Workstation, autosampler control included | |
Software | ClarityChrom 8.1 - PDA extension |
Preparative system configuration
Instrument | Description | Article No. |
Pump | AZUAR P2.1L, | |
Ternary LGP module | AZURA LPG ternary module for Pump P 2.1L | |
Assistant | AZURA ASM 2.2L Left: V4.1 + 6Port2Pos, 1/16”, sst, 500 bar Middle: UVD 2.1S Right: V 4.1 + 8Mpos, 1/8”, sst, 200 bar | - |
Flow cell | 3 µl; 1/16” | |
Dynamic mixer | Preparative HPLC, sst, 1/8”, 250V | |
Column | Eurospher II 100-10 C18, Column 150x50 mm | |
Software | PurityChrom®Basic |
References
[1] Bonini, S. A. et al. Cannabis sativa: A comprehensive ethnopharmacological review of a medicinal plant with a long history. Journal of Ethnopharmacology 227:300-315 (2018).
[2] Amin, M. R., Ali, D. W. Pharmacology of Medical Cannabis. Advances in Experimental Medicin and Biology 1162:151-165 (2019).
[3] ElSohly, M. A. et al. Phytochemistry of Cannabis sativa L. Progress in the Chemistry of Organic Natural Products 103:1-36 (2017).
[4] Lafaye, G., Karila, L., Blecha, L., Benyamina, A. Cannabis, cannabinoids, and health. Dialogues in Clinical Neuroscience 19(3): 309–316 (2017).
[5] Pisanti, S. et al. Cannabidiol: State of the art and new challenges for therapeutic applications. Pharmacology & Therapeutics 175:133-150 (2017).
[6] Loxterkamp, L., Monks, K. (C)an(n)alyze: determination of 16 cannabinoids inside flowers, oils and seeds. KNAUER Wisschenschaftliche Geräte GmbH (2020).
Related KNAUER Applications
VPH0070 – (C)an(n)alyze: determination of 16 cannabinoids inside flowers, oils and seeds
VPH0072 – Analyzing cannabis flowers according to the German Pharmacopeia - monograph 2018
Application details
Method | HPLC |
Mode | RP |
Substances | Cannabinoids |
CAS number | 24274-48-4, 2808-33-5, 13956-29-1, 521-35-7, 1972-08-3, 21366-63-2, 20675-51-8 |
Version | Application No.: VPH0073 Version 1 06/2020 | ©KNAUER Wissenschaftliche Geräte GmbH |



